Differentiating Viral from Bacterial Infections
Differentiating Viral from Bacterial Infections
Estimates claim that over 1.2 million people died in 2019 as a direct result of an antibiotic-resistant bacterial infection. Statistics show that up to 4.95 million deaths in the same year were associated with antimicrobial resistance (AMR)1. The overuse and misuse of antibiotics is considered to be the largest contributing factor to the rise of AMR. Antibiotics are effective at treating a wide range of bacterial infections, however, when used to treat viral infections, they have little to no effect. Even still, many physicians continue to prescribe so-called empirical antibiotics as an all-encompassing treatment strategy. In their defence, differentiating viral from bacterial infections can be troublesome. Traditional testing takes the form of paired serology, which requires patients to visit a healthcare facility twice during a 2–4-week period. Many of these infections have distressing symptoms, making this an unreasonable time-to-diagnosis period. Novel molecular techniques can reduce the time to result in the determination of many infections. However, some of these methods are associated with high false positive rates and low specificity resulting in further misuse of antibiotics.
Mxyovirus resistance protein A (MxA) is a biomarker associated with viral infections. It displays antiviral activity against positive, double-stranded RNA viruses and some DNA viruses2. In a study from earlier this year, MxA was used to differentiate viral from bacterial infections in a cohort of 61 adults with an AUROC of 0.9 and a sensitivity and specificity of 92.3% and 84.6% respectively3. An additional study, known as the TREND study, found that a cut-off of 430μg/L could effectively differentiate bacterial and viral infections with an AUROC of 0.9, a sensitivity of 92% and a specificity of 100%4.
C-reactive protein (CRP) is a non-specific acute phase protein which is associated with bacterial infection. However, CRP levels have also been shown to be elevated in response to various viral infections such as Influenza virus, malaria5 and SARS-COV-26, limiting its utility in differentiating the aetiology of an infection.
Using both biomarkers in combination can help physicians determine the true aetiology of infection with high specificity, supporting antimicrobial stewardship and reducing the harmful use of these drugs. Available on the VeraSTAT, Randox provides tests for MxA and CRP, which together provide a fast and accurate method of detection and differentiation of bacterial and viral infections from a small sample.
Alternatively, don’t hesitate to browse our range on our website or get in touch with one of our team at marketing@randox.com who will be happy to help with any query you have!
References
- Murray CJL, Ikuta KS, Sharara F, et al. Global burden of bacterial antimicrobial resistance in 2019: a systematic analysis. The Lancet. 2022;399(10325):629-655. doi:10.1016/S0140-6736(21)02724-0
- Liao S, Gao S. MxA: a broadly acting effector of interferon-induced human innate immunity. Visualized Cancer Medicine. 2022;3:2. doi:10.1051/vcm/2022002
- Metz M, Gualdoni GA, Winkler HM, et al. MxA for differentiating viral and bacterial infections in adults: a prospective, exploratory study. Infection. Published online February 3, 2023. doi:10.1007/s15010-023-01986-0
- Rhedin S, Eklundh A, Ryd-Rinder M, et al. Myxovirus resistance protein A for discriminating between viral and bacterial lower respiratory tract infections in children – The TREND study. Clinical Microbiology and Infection. 2022;28(9):1251-1257. doi:10.1016/j.cmi.2022.05.008
- Joseph P, Godofsky E. Outpatient Antibiotic Stewardship: A Growing Frontier—Combining Myxovirus Resistance Protein A With Other Biomarkers to Improve Antibiotic Use. Open Forum Infect Dis. 2018;5(2). doi:10.1093/ofid/ofy024
- Paranga TG, Pavel-Tanasa M, Constantinescu D, et al. Comparison of C-reactive protein with distinct hyperinflammatory biomarkers in association with COVID-19 severity, mortality and SARS-CoV-2 variants. Front Immunol. 2023;14. doi:10.3389/fimmu.2023.1213246
RX Imola: Inflammatory Biomarkers in COVID-19
Over the course of human history, few events have had such a dramatic impact as the COVID-19 pandemic. According to the World Health Organization (WHO), as of 12th July 2023, the SARS-CoV-2 virus has claimed almost 7 million lives and figures continue to rise1. While many who become infected are only subject to mild symptoms, those who develop a more severe form of the infection are encumbered with a debilitating flu-like condition, often requiring days, if not weeks of bed rest. In a paper from June 20232, the Rx Imola was used to study C reactive protein concentrations, along with other biomarkers, in mild and severe COVID-19 patients in order to develop novel risk stratification methods for this potentially life-threatening viral infection.
The impact on healthcare services around the world cannot be understated. In developed countries, access to services for both COVID-related and other conditions took a catastrophic hit. In low-to-middle-income countries, the impact has been even more distressing, all but eliminating basic medical care in favour of combating COVID-19, partly due to inferior resources and facilities3.
In times of medical emergency, it is crucial to have an efficient and effective means of stratifying the risk to patients and a process for suitably categorising those into the least and most at risk of severe complications or death. Due to the rate at which COVID-19 spread, unfortunately, the world lacked these mechanisms for SARS-CoV-2, resulting in mass hospital overpopulation, cancelled appointments for other life-threatening conditions and ultimately the staggering mortality statistics we’ve been bombarded with since January 2022. This prompted an unprecedented surge in medical research and major advances in testing capabilities, giving us new methods of detecting SARS-CoV-2 and determining the risk posed to individuals.
One such investigation, by Paranga et al., (2023) studied a total of 13 biomarkers to determine which could accurately differentiate mild, moderate, and severe cases and identify biomarkers which were good predictors of fatality2. C reactive protein (CRP) was the best-described biomarker relating to COVID-19 throughout the pandemic. This paper compares it to 12 other biomarkers including suPAR, sTREM-1, ferritin, MCP-1 and Lactate dehydrogenase. Of these, it was discovered that CRP was clearly the most effective biomarker for differentiating mild from severe cases, with concentrations in those with severe infection being, on average, 45% higher than in those with mild symptoms2. Additionally, the authors discovered that CRP levels were not significantly affected by age, a factor known to affect the inflammation and immune responses, providing a powerful and inclusive risk stratification tool. Some of the additional conclusions drawn from this paper can be seen below2:
- Lactate dehydrogenase, sTREM-1 and HGF were good predictors of mortality in COVID-19.
- suPAR was identified as a crucial molecule in characterising Delta variant infection and mortality.
- The initial values of inflammatory biomarkers were good to excellent predictors of disease severity in COVID-19 patients.
- Disease severity and mortality are associated with a higher rate of comorbidities including thrombocytopenia and other blood diseases, circulatory and respiratory system diseases and liver diseases such as cirrhosis.
So, what is CRP and how does it become elevated in response to a SARS-CoV-2 infection? CRP is a non-specific, acute-phase protein, meaning its concentration is altered in response to inflammation4. The acute respiratory distress syndrome induced by SARS-CoV-2 is, in part, a result of the hyperinflammation caused by the virus2. CRP is a well-characterised inflammatory biomarker and is therefore well-suited for identification and risk stratification in an emerging disease.
This investigation2 utilised the RX Imola, a rapid, comprehensive clinical chemistry platform, to quantify CRP. With the RX Imola, laboratories can gain access to the world’s largest clinical chemistry test menu covering routine chemistries as well as specific proteins, lipids, and more providing a cost-effective and user-friendly platform. With 60 cooled reagent positions and a sample carousel with 20 cooled positions for controls and calibrators, the RX Imola is an ideal solution for small to medium-throughput laboratories seeking an innovative and reliable clinical chemistry system. Randox also supplies suitable, high-quality reagents, and through Acusera, state-of-the-art controls and calibrators, completing the clinical chemistry portfolio.
References
1. World Health Organisation. WHO Coronavirus (COVID-19) Dashboard. https://covid19.who.int/.
2. Paranga TG, Pavel-Tanasa M, Constantinescu D, et al. Comparison of C-reactive protein with distinct hyperinflammatory biomarkers in association with COVID-19 severity, mortality and SARS-CoV-2 variants. Front Immunol. 2023;14. doi:10.3389/fimmu.2023.1213246
3. Jain P. Impact of COVID-19 Pandemic on Global Healthcare Systems and the role of a new era of global collaborations. Sushruta Journal of Health Policy & Opinion. 2021;14(3):1-5. doi:10.38192/14.3.2
4. Nehring S. C Reactive Protein . https://www.statpearls.com/articlelibrary/viewarticle/18744/.
Identification and Differentiation of Viral and Bacterial Respiratory Infection to Guide Antibiotic Stewardship

Identification and Differentiation of Viral and Bacterial Respiratory Infection to Guide Antibiotic Stewardship
The development of point-of-care testing is critical in the identification and differentiation between bacterial and viral respiratory infections. Defining the indications of infection to improve antibiotic stewardship, ensures that patients are protected from unnecessary antibiotic use and antibiotic resistance. It has been shown that particular protein biomarkers, such as myxovirus resistance protein (MxA) and C-reactive protein (CRP), differentiate infections between bacterial and viral. Using point-of-care platforms, such as Randox’s VeraSTAT, for detection of these protein biomarkers may provide more rapid and cost-effective discriminating tools.
The treatment of bacterial and viral infections can differ significantly, however people are often treated with empirical antibiotics due to a lack of paid and accurate testing. Although early intervention of infection is urgent, current diagnostic methods are either time intensive or inaccurate. The challenges clinicians are faced with in the differentiation of viral or bacterial respiratory infection can lead to delayed diagnosis, misappropriation of antibiotics and increased healthcare costs.
MxA protein has the potential to greatly enhance the rapid detection of viral respiratory infections as it increases significantly when there is actuate viral infection. CRP is the dominant acute phase protein often used to guide treatment of a bacterial infection or inflammation associated with tissue injury, inflammatory disorders, and associated diseases.

CRP & MxA together, allow clinicians to make appropriate decisions in supporting antimicrobial stewardship and guide the appropriate use of antibiotics, saving time performing unnecessary tests, providing unnecessary treatment which missing the opportunity to provide the right treatment in a timely manner.
The Randox VeraSTAT is a simple, accurate, portable point of care device which delivers rapid results via the use of patented cathodic electrochemiluminescence technology (C-ECL). Designed with the aim of offering users the next generation of rapid diagnosis, the VeraSTAT eliminates the requirement to send samples to a laboratory and instead returns results in as little as 6 minutes.
- Eliminates delays in sending samples to the lab and facilitate immediate decision making at the point of care.
- Lightweight, portable and convenient, the Randox VeraSTAT can be used in a variety of locations to deliver results as required, such as a GP surgery or Emergency Department.
- Intuitive user interface guides the operator through the entire testing process.
- All necessary reagents are conveniently included in each single use, sealed cassette with no preparation required. All necessary consumables are supplied with the kit.
- The Randox VeraSTAT allows for results to be exported via Bluetooth connectivity.
- Flexible test menu comprising of a range of immunoassay, protein, inflammatory, diabetes & infectious disease markers.
Novel testing approaches identifying the type of infection at the point of care are essential in accurately guiding appropriate antibiotic treatment. Although these tests can’t determine what type of viral or bacterial infection a patient has, it will determine whether the infection is viral or bacterial, further testing is then carried out to determine what type of pathogen the patient has via PCR – the gold standard. The ability to distinguish between viral and bacterial infections is the most effective guide for clinical decision making and is an innovative tool for antibiotic stewardship.
References
1 – Fleming-Dutra K.E., Hersh A.L., Shapiro D.J. Prevalence of inappropriate antibiotic prescriptions among US ambulatory care visits, 2010–2011. JAMA. 2016;315:1864–1873. doi: 10.1001/jama.2016.4151.
2 – Cals JW, Hopstaken RM, Butler CC, Hood K, Severens JL, Dinant GJ. Improving management of patients with acute cough by C-reactive protein point of care testing and communication training (IMPAC3T): study protocol of a cluster randomised controlled trial. BMC Fam Pract. 2007;8:15.
3- New report calls for urgent action to avert antimicrobial resistance crisis [Internet]. World Health Organization. World Health Organization; 2019
4 – Hutchings MI, Truman AW, Wilkinson B. Antibiotics: past, present and future. Curr Opin Microbiol. (2019) 51:72–80. doi: 10.1016/j.mib.2019.10.008
For more enquiries please contact the Marketing team: market@randox.com
ABOUT RANDOX
NEWS
VACANCIES
OUR PEOPLE
CRP Assay
Reagent | C-Reactive Protein (CRP)
C-Reactive Protein (CRP): The Most Sensitive Acute Phase Reactant
Benefits of the Randox Assay


Limited Interference
The Randox CRP assay has shown to have limited interference from conjugate and free bilirubin, haemoglobin, Intralipid® and triglycerides.


Liquid ready-to-use
The Randox CRP assay is available in a liquid ready-to-use format for convenience and ease-of-use.


Stable to expiry
The Randox CRP assay is stable to expiry when stored at +2oC to +8oC.


Applications available
Applications available detailing instrument-specific settings for the convenient use of the Randox CRP assay on a variety of clinical chemistry analysers.


Dedicated calibrator and controls available
Dedicated CRP calibrator and controls available offering a complete testing package.
Ordering Information
| Cat No | Size | ||||
|---|---|---|---|---|---|
| CP7950 | R1 7 x 20ml (L) R2 2 x 12ml | Enquire | Kit Insert Request | MSDS | Buy Online |
| CP9742 | R1 6 x 66ml (L) R2 6 x 13ml | Enquire | Kit Insert Request | MSDS | Buy Online |
| CP3826 | R1 6 x 20ml (L) R2 3 x 9ml | Enquire | Kit Insert Request | MSDS | Buy Online |
| (L) Indicates liquid option | |||||
Instrument Specific Applications (ISA’s) are available for a wide range of biochemistry analysers. Contact us to enquire about your specific analyser.
More Information
C-reactive protein (CRP) is the dominant acute phase protein in mammals 1. Elevated levels of CRP are not disease specific but are synthesised, by hepatocytes, in response to pro-inflammatory cytokines during inflammatory and/or infectious processes 2, 3. The physiological function of CRP is the enhancement of cell-mediated immunity through the promotion of phagocytosis, accelerated chemotaxis and the activation of platelets 1.
Known as a biomarker of acute inflammation, many large-scale prospective studies demonstrate the association between CRP and chronic inflammation, including: cardiovascular disease (CVD), cerebrovascular accident (CVA) (ischaemic stroke), Alzheimer’s Disease, and age-related macular degeneration 2.
CRP is a major cardiovascular disease (CVD) risk factor. A CRP level of >10mg/l correlates with a >4% risk of developing a fatal CVD event in 10 years, including: myocardial infarction, peripheral arterial disease, stroke and sudden cardiac death. Inflammation is an integral part in the development and rapid progression of coronary heart disease (CHD) 4.
The coronary artery disease (CAD) process is characterised by increasing levels of inflammatory biomarkers. CRP is not only an excellent biomarker or mediator of atherosclerosis but is a strong independent marker in the prediction of adverse CV events, including: myocardial infarction, ischaemic stroke, and sudden cardiac death. CPR can be utilised as a clinical guide for the diagnosis, management and prognosis of CVD 5.
The development of a cerebrovascular accident (CVA) (stroke) is the result of longstanding vascular inflammation, thrombosis, plaque rupture and subsequent brain ischaemia or infarction. Elevated CRP levels is associated with the development of CVA 6.
High sensitivity CRP (hsCRP) plays an important role in the early diagnosis, prognosis, long and short death risk and prediction of etiological subtypes of stroke 7. Find out more about the Randox hsCRP assay.
Alzheimer’s disease (AD) is the most common form of dementia, accounting for 60-80% of dementia cases 7. CRP is believed to be involved in the pathophysiology of cerebral small vessel disease (CSVD) and neurodegeneration 8. CRP levels are associated with cognitive impairment 9. Inflammation should be considered as a target treatment, with the aim of delaying the progression of subclinical brain damage and cognitive decline 8. Midlife elevations in CRP are associated with an increased risk of developing AD. Patients with AD, CRP elevations continues to predict increased dementia severity suggestive of a possible pro-inflammatory endophenotype in AD 10.
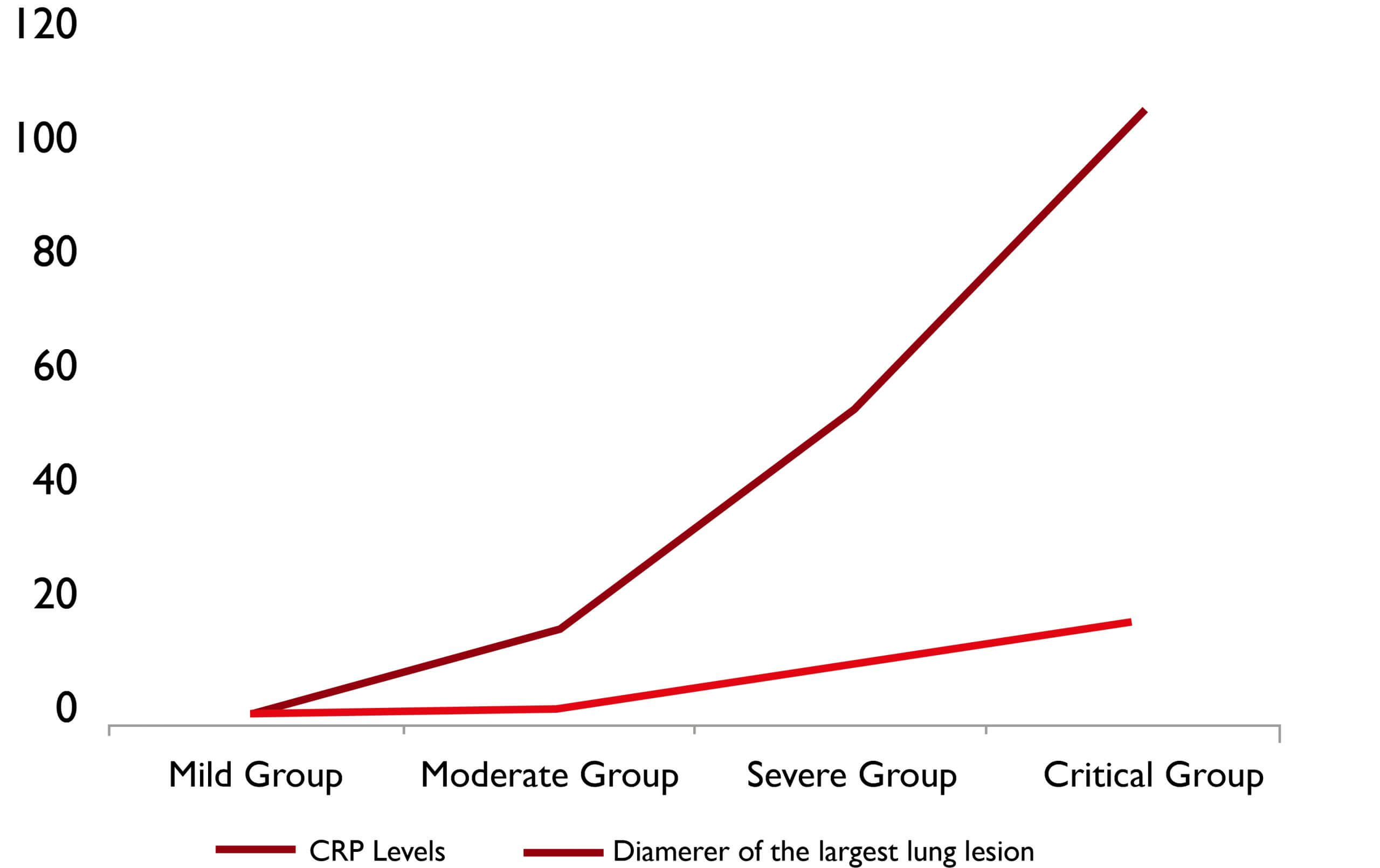
In COVID-19 patients, CRP testing has proved to perform well in discriminating disease severity and predicting adverse outcomes 11. CRP levels positively correlates with lung lesions, reflecting disease severity, and should be considered a key indicator in disease monitoring (see fig. 1) 12.
CRP levels are associated with computed tomography (CT) scores and COVID-19 disease development, with CRP levels increasing in the initial stage of severe COVID-19, prior to the CT findings 13.
Elevated CRP levels have been identified in 86% of patients admitted to hospital. CRP measurements are useful in diagnosis, assessing prognosis and monitoring for clinical improvements or deterioration 14.
Fig. 1. Relation between CRP levels, clinical severity, and lung lesions 12

CRP Calibrator
CRP Control
Specific Protein EQA
Reagents Homepage
References
Rheumatoid Arthritis and Women’s Health
Rheumatoid Arthritis (RA) is a chronic autoimmune disease characterised by pain, swelling and stiffness in joints which commonly affects the hands, wrists and feet. Whilst both men and women can suffer from rheumatoid arthritis, it is more commonly seen in women than men.
Rheumatoid arthritis is the most common autoimmune disease with a higher prevalence rate compared to lupus, multiple sclerosis, type 1 diabetes, Crohn’s disease and psoriasis.


(Simmons, 2013)
The incidence rates of rheumatoid arthritis differ between men and women. The onset of RA occurs much earlier for women, for most, during their 30’s and 40’s. In an American study, it was noted that the incidence rates peak for women around the ages of 55 to 64, compared to 75 to 84 years of age for men.


(Simmons, 2013)
As most women are diagnosed with rheumatoid arthritis in their 30’s and 40’s, a study found that the diagnosis negatively impacts both the body and mind of women, as indicated in their pain, disease activity, and quality of life scores. This is due to women being diagnosed at a time when their burdens are the heaviest as this is the time when women are most likely to have children or are raising children combined with work and socialising.
Changes in hormone levels also impacts women. It has been noted that before a menstrual period, women find the symptoms of rheumatoid arthritis to be more severe, but settles during their cycle. Also, due to the changes in hormone levels during pregnancy, 50 – 60% of women with rheumatoid arthritis noticed that their symptoms improved.
The key to managing rheumatoid arthritis is to start the treatment as early as possible as it can halt or slow the disease, preventing joint damage and complications, including: osteoporosis and cardiovascular disease. Rheumatoid arthritis increases the risk of heart attack by 60%. To start treatment as early as possible, it is important that it is diagnosed as early as possible.
Randox offer a number of key assays for the diagnosis of rheumatoid arthritis.
Rheumatoid factor is the most routinely run test to diagnose rheumatoid arthritis as 80% of rheumatoid arthritis patients test positive for rheumatoid factor. The Randox Rheumatoid Factor reagent offers the following benefits:
- Wide measuring range of 6.72 – 104lU/ml for the accurate measurement of clinically important results
- Accurate assessment of rheumatoid factor titre (calibrant standardised against primary WHO material; 1st British Standard 64/2)
- No interference from complement C1q
- Automated immunoturbidimetric assay
- Applications available for a wide range of biochemistry analysers, detailing instrument-specific settings
It has been found that complement C4 and CRP upregulation indicates the middle to late stages of rheumatoid arthritis.
The Randox Complement C4 reagent offers the following benefits:
- Wide measuring of 3.41 – 152mg/dl for the accurate measurement of clinically significant results
- Limited interferences from Bilirubin, Haemoglobin, Intralipids, and Triglycerides, producing more accurate results
- Automated immunoturbidimetric assay
- Applications available for a wide range of biochemistry analysers, detailing instrument-specific settings
The Randox High-Sensitivity CRP reagent offers the following benefits:
- Wide measuring of 0.477 – 10mg/l fir the accurate measurement of clinically significant results
- Liquid ready-to-use reagents for convenience and ease of use
- Applications available for a wide range of biochemistry analysers, detailing instrument-specific settings


Neonatal health testing from Randox: keeping your baby healthy now and into the future
Most newborns enter the world healthy. But sometimes, infants develop conditions that require medical tests and treatment. Newborns are particularly at risk for some diseases, and in particular infections, because their immune systems aren’t developed enough to fight bacteria, viruses, and parasites.
At Randox we offer a number of accurate and reliable tests capable of detecting illnesses in newborn babies, enabling early medical intervention to allow for the best possible outcome for the baby.
Testing for Jaundice with Randox Bilirubin
In the routine care of newborns, a test for bilirubin is commonly conducted.
Bilirubin is formed by the breakdown of haemoglobin in the spleen, liver and bone marrow. It travels to the liver where it is secreted into the bile ducts as bile, and stored in the gallbladder where it is later released into the small intestines for digestion.
Increased levels of bilirubin within the body are associated with a condition called jaundice, which occurs in toxic or infectious diseases of the liver. The most common symptom of jaundice is a yellow pigmentation of the skin.
Elevated levels of bilirubin may also arise as a result of an obstruction in the bile duct or gall bladder, as a result of haemolysis (the destruction of red blood cells), or by the liver not actively treating the haemoglobin it is receiving.
Therefore the Randox Bilirubin test is essential in the screening, monitoring and diagnosis of hepatic (liver function) disorders and jaundice in newborn babies.
Neonatal jaundice, otherwise known as hyperbilirubinemia, is extremely common in babies, because nearly every newborn develops a somewhat elevated bilirubin level during the first week of life.
Side effects may include excess sleepiness or poor feeding, but in some more extreme cases babies may experience seizures, cerebral palsy, delayed intellectual development, or physical abnormalities.
Early and accurate detection is therefore extremely important – making bilirubin testing fundamental. To ensure the precision of the bilirubin tests conducted in paediatric testing, Randox also offers Acusera Bilirubin Elevated Quality Control.
Monitoring the destruction of red blood cells with Randox G-6-PDH
Glucose-6-Phosphate Dehydrogenase (G-6-PDH) is an enzyme located on the X-chromosome, and so is found in every bodily cell as soon as a baby is born.
G-6-PDH is involved in the normal processing of carbohydrates and plays a critical role in red blood cells, protecting them from damage and destruction. Depleted levels of G-6-PDH can therefore cause red blood cells to become particularly vulnerable to haemolysis. G-6-PDH deficiency, which causes rapid heart rate, shortness of breath, excess tiredness, and mild to severe jaundice in new-borns, affects more than 400 million people globally.
During a baby’s new-born screening, a test for the G-6-PDH enzyme will be conducted to check for this deficiency disorder. Early diagnosis is imperative, as untreated haemolysis can result in haemolytic anaemia.
Genetic Disease Screening with Randox Copper
Copper is an essential mineral in human nutrition, and is mainly found in the brain, liver, kidneys, heart and skeletal muscle.
It aids in some of the key bodily functions including the production of red blood cells, the maintenance of nerve cells and the immune system, and the formation of bone and connective tissue. A deficiency in this mineral can therefore result in bone abnormalities or fractures in premature babies.
Copper deficiency can also be caused by an inherited disorder called Menkes Disease. Affecting approximately 1 in 100,000 children worldwide, this condition is characterised by sparse, kinky hair; failure to gain weight and grow at the expected rate, and deterioration of the nervous system.
The first signs of Menkes Disease – curly, sparse, coarse, dull, and discoloured hair – usually first develop at 2-3 months of age and therefore monitoring copper levels in babies is a way to catch this rare condition at the earliest possible opportunity.
Testing for Lupus with Randox Complement C4 and Complement C3
Another condition which can affect newborn babies is neonatal lupus, which occurs when the mother’s antibodies affect the foetus. A rare condition, it is an autoimmune disease caused by the body’s immune system attacking its own tissues and organs.
The Complement C4 and Complement C3 proteins, which play an important role in eliminating certain infections, can be used as biomarkers in the diagnosis and monitoring of lupus. Complement C4 deficiency is commonly associated with lupus, as the protein is required to clear damaged cells, promote inflammation, and attack pathogens.
Although there is no cure for lupus at present, the condition is very treatable and usually responds well to a number of different types of medication – especially when treatment is started in the early stages of the disease.
Early diagnosis is therefore imperative, and the Randox Complement C4 and Complement C3 tests can help to diagnose babies with lupus at the earliest possible stage. Randox also offer Acusera Immunology controls.
Monitoring a baby’s anti-infection defences with Randox IgA
IgA (immunoglobulin A) is an antibody present in the cells of the immune system, and plays a crucial role in the immune function of mucous membranes including tears, saliva, and sweat. It is also present in colostrum, often referred to as ‘liquid gold’, which is the first secretion from the mammary glands after giving birth.
It’s the IgA in colostrum and milk that is important in neonatal protection against infection and it is therefore imperative to monitor the levels of this antibody to make sure your baby is receiving the anti-infection defences he or she requires.
Testing for allergic reactions with Randox IgE
IgE (immunoglobulin E) is an antibody released by the immune system as a defence mechanism when it believes the body is at risk. IgE determinations are therefore used as an aid in the diagnosis of allergic diseases.
In babies, an allergen-specific IgE test may be done to look for some kinds of allergies, including food, animal dander, pollen, mould, medicine, dust mites, or insect venom.
Increased concentrations of IgE will confirm that an allergic response has occurred, facilitating further investigation as to the specific allergy present.
Testing for bacterial infection with Randox CRP
C-reactive protein (CRP) is an acute phase protein found in blood plasma and produced by the liver. The concentration levels of CRP increase in response to cytokines which are produced by white blood cells during inflammation, infection and tissue injury.
Testing for this protein can therefore be used in the detection of bacterial infections in neonates – enabling antibiotic prescription and a speedy recovery. If infection is identified, CRP can also be used to monitor treatment response or identify neonatal septicaemia.
Randox is committed to saving and improving lives – at any age and any stage of life.
Our innovative diagnostic technologies are versatile and easily adapted for use in the paediatric setting – keeping your baby healthy now and into the future.
For more information on neonatal health tests available from Randox click here or email randoxpr@randox.com or phone 028 9442 2413
Inflammatory Biomarker Series: CRP
An inflammatory biomarker detects inflammation in the body. Inflammation is not just the immediate, short-term response of the body to an injury or infection. Inflammation within the body can be a long-term, chronic condition resulting in a number of health implications. In diagnostics, measurement of an inflammatory biomarker can not only detect acute inflammation but provide a marker of treatment response.
C-reactive protein (CRP) is an acute phase protein produced by the liver in response to inflammation, infection and tissue injury. CRP is a particularly beneficial inflammatory biomarker as it is detected much faster than other markers in the blood. Levels of CRP increase when inflammation occurs and therefore it can be a significant biomarker in a range of diseases, including the following.
Cardiovascular Disease
An increasing amount of research exists to suggest CRP is not only a useful, non-specific inflammatory biomarker, but it may have a direct influence on coronary heart disease and cardiac events1. Inflammation can occur when LDL cholesterol builds up in the artery walls causing atherosclerosis. Modifiable risk factors of atherosclerosis include smoking, diabetes, poor diet, high blood pressure and physical inactivity, all factors which subsequently increase the risk of heart attacks, ischemic stroke, peripheral artery disease and even vascular dementia2,3.
Studies have also shown that persistent low levels of CRP can contribute to a person developing CVD. Therefore using high sensitivity CRP as an inflammatory biomarker can detect low levels, helping to predict the likelihood of a patient developing CVD in the future.
Diabetes
Research suggests that inflammation in the body can influence the development of type 2 diabetes. With the ability to be managed through diet and exercise, type 2 diabetes is commonly associated with obesity. Research has shown that excess body fat can cause continuous chronic low-grade inflammation as a result of inflammatory cytokines and increased plasma levels of CRP. As a result, this chronic inflammation has the ability to cause insulin resistance leading to the development of type 2 diabetes4.
Rheumatoid Arthritis
A three year study which analysed the bone and joint health of 10,000 patient samples in India has found that inflammatory biomarkers, in particular CRP and ESR (Erythrocyte Sedimentation Rate) were raised in most of the samples compared to any other markers5. Although CRP is a non-specific inflammatory biomarker, it can be used alongside other tests, such as Rheumatoid Factor, to diagnose inflammatory joint diseases such as Rheumatoid Arthritis. Not only will CRP levels be higher due to chronic inflammation, but CRP levels can be monitored to assess levels of inflammation over time, allowing clinicians offer effective treatment.
Chronic Obstructive Pulmonary Disease (COPD)
COPD is a condition associated with inflammation of the lungs and airways. Studies have shown that measuring CRP levels is beneficial to detect exacerbations, when symptoms of COPD get suddenly worse and can last for several days. This is because CRP levels spike when exacerbations happen, causing lung function to deteriorate6.
Neonatal Bacterial Infections
CRP is one of the preferred and frequently used tests in neonatal units when diagnosing suspected bacterial infections, such as neonatal sepsis, in newborns who show signs on infection. Due to delayed synthesis during the inflammatory response, the sensitivity of CRP is lowest during early stages of infection. It is therefore critical that extremely low levels of CRP can be detected during diagnosis to distinguish whether symptoms are related to an infectious or non-infectious condition. This early detection then allows for rapid and appropriate neonatal treatment7.
Inflammatory Bowel Disease
Research suggests that using CRP as an inflammatory biomarker can help distinguish between Inflammatory Bowel Disorder (IBD) and Irritable Bowel Syndrome (IBS)8. Although IBD and IBS have some similarities in symptoms, IBD causes chronic inflammation, whereas IBS is a non-inflammatory condition. Therefore using CRP as a biomarker can allow clinicians to deliver a confident and accurate diagnosis.
For health professionals
Randox Laboratories manufacture a wide range routine and niche biochemistry reagents for use in both a research and clinical setting. With a wide measuring range, the Randox CRP assay will perform excellently to detect levels outside of the healthy range. Also available is a Full Range CRP assay particularly beneficial for use in a neonatal setting, and a High Sensitivity CRP assay, depending on your diagnostic requirements. For more information, please contact: reagents@randox.com
References:
- Shrivastava, A. K., Singh, H.V., Raizada, A. and Singh, S.K. C-reactive protein, inflammation and coronary heart disease. The Egyptian Heart Journal. 67, 89-97. (2015)
- American Heart Association. Inflammation and Heart Disease. Available from: https://goo.gl/d82Ynr (2016)
- Harvard Health Publications. What you eat can fuel or cool inflammation. Harvard Health Publications. Available from: https://goo.gl/e8m3El (2007)
- Zeyda, M. and Stulnig, T. M. Obesity, Inflammation, and Insulin Resistance – A Mini-Review. Gerontology 2009; 55:379-386 (2009)
- Mukherjeel, R. Bone and joint health are crucial aspect, usually ignored by Indians. The Times of India. Available from: https://goo.gl/qluzhI (2016)
- Anderson, G. P. COPD, asthma and C-reactive protein. European Respiratory Journal 2006; 27: 874-876. (2006)
- Hofer, N., Zacharias, E., Müller, W. and Resch, B. An update on the Use of C-Reactive Protein in Early-Onset Neonatal Sepsis: Current Insights and New Tasks. Neonatology 2012; 102: 25-36 (2012)
- Silva, P. Two Specific Proteins Allow the Exclusion of IBD in Patients with Irritable Bowel Syndrome. IBD News Today. Available from: https://goo.gl/pxMP53 (2015)


Immunology / Protein Control Calibrators




β-2-Microglobulin Calibrator
Dedicated β-2-Microglobulin calibrators designed for use in the calibration of b-2-Microglobulin assays. With an excellent working stability of 30 days at 2ºC to 8ºC, waste is kept to a minimum.
Features & Benefits
- Lyophilised for enhanced stability
- 100% human serum
- Stable for 30 days at +2-8°C
| Description | Size | Analytes | Cat No | |
|---|---|---|---|---|
| β-2Microglobulin Calibrator | 3 x 1ml | 1 | BM10444 | |
| β-2Microglobulin Calibrator | 3 x 1ml | 1 | BM1362 | |
Analytes
- β-2 Microglobulin
CRP Calibrator
Dedicated CRP calibrator designed for use in the calibration of CRP assays. Calibrators are available to suit high sensitivity, mid range and full range assays.
Features & Benefits
- Liquid ready-to-use
- 100% human serum
- Stable for 30 days at +2-8°C
| Description | Size | Analytes | Cat No | |
|---|---|---|---|---|
| CRP High Sensitivity Calibrator Series | 6 x 2ml | 1 | CP2478 | |
| CRP Full Range Calibrator Series (0.1-160mg/l) | 6 x 2ml | 1 | CP2499 | |
Analytes
- CRP
Cystatin C Calibrator
Dedicated Cystatin C calibrator designed specifically for use with the Randox Cystatin C assay. Five different levels are provided in a liquid stable format for ease of use and convenience.
Features & Benefits
- Liquid ready-to-use
- 100% human serum
- Stable to expiry date at +2-8°C
- Open vial stability of 30 days at +2-8°C
- Multi-point calibrator
| Description | Size | Analytes | Cat No | |
|---|---|---|---|---|
| Cystatin C Calibrator | 5 x 2ml | 1 | CYS10445 | |
Analytes
- Cystatin C
IgE Calibrator
Dedicated IgE calibrator specifically designed for use with immunoturbidimetric IgG assays.
Features & Benefits
- Liquid ready-to-use
- 100% human serum
- Stable to expiry date at +2-8°C
- Open vial stability of 30 days at +2-8°C
| Cat No | Size | Analytes | Cat No | |
|---|---|---|---|---|
| IgE Calibrator Series | 6 x 1ml | 1 | IE2492 | |
Analytes
- lgE
Immunoglobulin Calibrator
Multi-analyte calibrator covering 3 immunoglobulin assays. Designed specifically for use with the Randox range of immunoglobulin assays.
Features & Benefits
- Liquid ready-to-use
- 100% human serum
- Stable to expiry date at +2-8°C
- Open vial stability of 30 days at +2-8°C
| Description | Size | Analytes | Cat No | |
|---|---|---|---|---|
| Immunoglobulin Calibrator | 3 x 1ml | 3 | IT3861 | |
Analytes
- IgA
- IgM
- lgG
Specific Protein Calibrator - For use with samples that do not require pre-dilution
Multi-analyte calibrator set covering 12 serum proteins. Five different levels are provided in a liquid stable format for ease of use and convenience.
Features & Benefits
- Liquid ready-to-use
- 100% human serum
- 5 levels provided
- Stable to expiry date at +2-8°C
- Open vial stability of 30 days at +2-8°C
| Description | Size | Analytes | Cat No | |
|---|---|---|---|---|
| Specific Protein Calibrator (Undiluted Samples) | 5 x 1ml | 12 | IT2691 | |
Analytes
- ASO
- Complement C3
- Complement C4
- CRP
- Ferritin
- Haptoglobin
- Prealbumin
- Rheumatoid Factor
- Transferrin
Specific Protein Calibrator - For use with samples that require pre-dilution
Multi-analyte calibrator set covering 3 serum proteins. Five different levels are provided in a liquid stable format for ease of use and convenience.
Features & Benefits
- Liquid ready-to-use
- 100% human serum
- 5 levels provided
- Stable to expiry date at +2-8°C
- Open vial stability of 30 days at +2-8°C
| Description | Size | Analytes | Cat No | |
|---|---|---|---|---|
| Specific Protein Calibrator (Diluted Samples) | 5 x 1ml | 3 | IT2692 | |
Analytes
- IgA
- IgG
- IgM
Soluble Transferrin Receptor (sTfR) Calibrator
Providing a true third party solution for the measurement of Soluble Transferrin Receptor (sTfR), the Acusera sTfR Calibrator will deliver an unbiased, independent assessment of analytical performance.
Features & Benefits
- Lyophilised for enhanced stability
- Human based serum
- Stable to expiry date at +2-8°C
- Open vial stability of 28 days at +2-8°C
| Description | Size | Analytes | Cat No | |
|---|---|---|---|---|
| sTfR Calibrator | 6 x 1ml | 1 | TF10161 | |
Analytes
- Soluble Transferrin Receptor (sTfR)
High Sensitivity CRP Quality Control




The Randox Acusera High Sensitivity CRP quality control is designed to monitor the performance of high sensitivity CRP assays. The liquid ready-to-use format makes it convenient and easy to use. Once opened the control is stable to expiry at +2-8°C significantly minimising waste and helping to reduce costs.
Features & Benefits
- Liquid ready-to-use
- 100% human serum
- Assayed target values provided
- Stable to expiry date at 2°C – 8°C
- Once opened stable to expiry date at 2°C – 8°C
| Description | Size | Analytes | Cat No | |
|---|---|---|---|---|
| High Sensitivity CRP Control Level 1 | 10 x 1ml | 1 | CP2476 | |
| High Sensitivity CRP Control Level 2 | 10 x 1ml | 1 | CP2477 | |
Analytes
- CRP
Related Products
CRP Quality Control




The Randox Acusera CRP quality control is designed to monitor the performance of traditional CRP assays. The liquid ready-to-use format makes it convenient and easy to use. Once opened the control is stable for 30 days at +2-8°C significantly minimising waste and helping to reduce costs.
Features & Benefits
- Liquid ready-to-use
- 100% human serum
- Assayed target values provided
- Stable for 30 days at 2°C – 8°C
| Description | Size | Analytes | Cat No | |
|---|---|---|---|---|
| Liquid CRP Control Level 2 | 10 x 1ml | 1 | CP2480 | |
| Liquid CRP Control Level 3 | 10 x 1ml | 1 | CP2481 | |
Analytes
- CRP





