COVID-19 – Laboratory Tools for Disease Management
In addition to tests for the detection of SARS-CoV-2, the virus that causes COVID-19, laboratory diagnostics plays a pivotal role in patient management, risk stratification, monitoring of disease progression, associated complications and response to treatment.
Randox offers a comprehensive range of laboratory solutions including; diagnostics reagents, revolutionary Biochip technology and quality control designed to provide clinicians with valuable insights into disease severity ultimately helping to improve patient care.
Patients with comorbidities such as diabetes mellitus, cardiovascular disease (CVD) and chronic kidney disease (CKD) are particularly susceptible to COVID-19 infection and are at greater risk of developing severe illness. The Randox portfolio may also be used to diagnose and monitor such at risk patients with underlying health concerns.
Diagnostic Solutions for COVID-19 Management and Monitoring
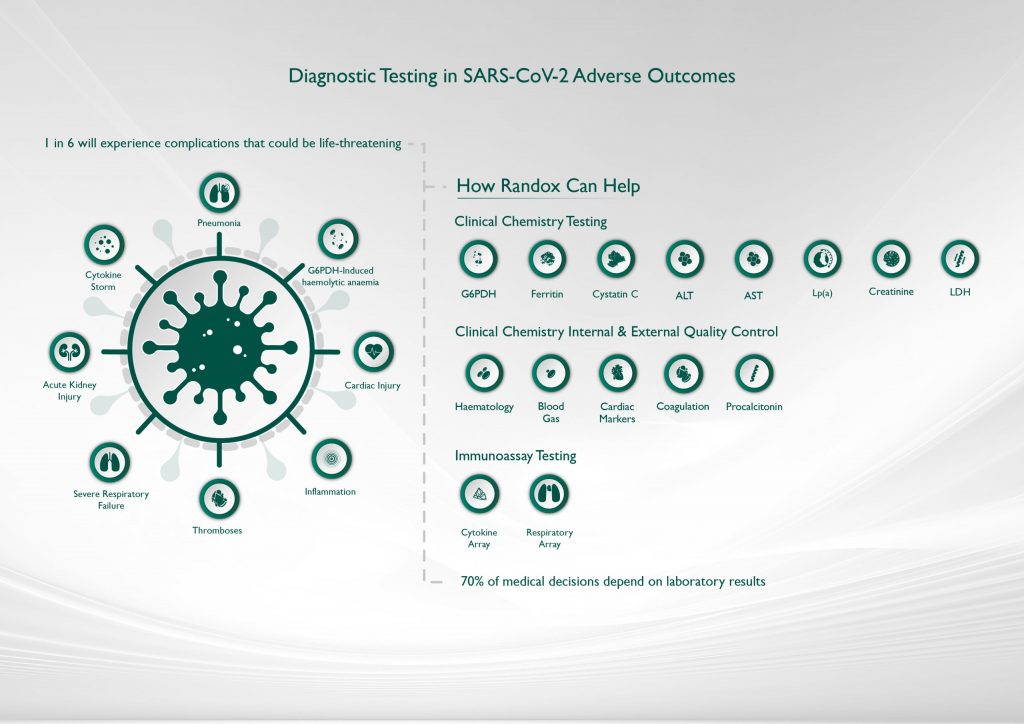
Our extensive test menu comprises 113 assays, including many unique tests and a range of biomarkers designed to assist laboratories when screening for a variety of COVID-19 related health issues.
Randox Reagents are internationally recognised as being of the highest quality; producing accurate and precise results. Applications are available detailing instrument-specific settings for the majority of clinical chemistry analysers.

C-Reactive Protein (CRP)
Elevated markers of inflammation and infection such as CRP may indicate severe viral infection. CRP is elevated in up to 86% of COVID-19 patients who are admitted to hospital. Testing for CRP is useful in assessing prognosis, and when monitoring improvement. It is recommended that CRP is measured at baseline and then at intervals, a sudden increase in levels will prompt surveillance for secondary infection or other cause of deterioration.

Ferritin
As an acute phase reactant, Ferritin levels increase in response to inflammation. High levels have been identified in approximately 60% of individuals hospitalised as a result of COVID-19 infection. In critically ill patients, elevated Ferritin levels may be indicative of a cytokine storm and secondary haemophagocytic lymphohistiocytosis, a hyperinflammatory syndrome associated with organ failure.

Liver Function Tests (Albumin, ALT, AST, Bilirubin)
Patients with COVID-19 may be at higher risk of liver injury or impairment of liver function. Liver function markers such as ALT, AST and Bilirubin are notably higher in more severe infections, decreased albumin levels have also been documented in severe cases. Drug induced liver injury as a result of COVID-19 treatment should not be ignored and should be monitored.

Renal Function Tests (Creatinine, Cystatin C)
Emerging evidence suggests that SARS-CoV-2 may directly affect kidney function, as such COVID-19 patients should be assessed for Acute Kidney Injury (AKI) on admission to hospital and monitored throughout their stay. AKI can lead to worse outcomes for patients.

Lactate Dehydrogenase (LDH)
LDH levels may be a convenient marker for the identification of COVID-19 patients at higher risk of developing severe respiratory failure. It is well documented that high LDH levels are indicative of haematological malignancy and acute lung injury. LDH levels might therefore reflect tissue necrosis related to immune hyperactivity and thus are related to poor clinical outcomes in individuals with COVID-19.

G6PDH
Chloroquine has been investigated as a potential treatment for COVID-19, the anti-malaria drug however could have devastating consequences for those with a G6PDH deficiency. G6PDH deficiency is a genetic disorder affecting the red blood cells, causing them to break down prematurely. The most common complication of deficiency is haemolytic anaemia. An estimated 400 million people globally have G6PDH deficiency, it is most common in certain parts of Africa, Asia, the Mediterranean and Middle East.

Lp(a)
Patients with either baseline elevated Lp(a) or patients whose Lp(a) levels increased following infection from COVID-19, or both, may be at a significantly increased risk of developing thromboses. Elevate Lp(a) levels may cause acute destabilisation of pre-existing but quiescent, atherosclerotic plaques, which could induce an acute myocardial infarction and stroke
Randox Biochip technology is extremely versatile with applications in not only the diagnosis of COVID-19 but also the identification of individuals at risk of developing more severe illness.
Unique to Randox, Biochip technology is capable of simultaneous detection of a panel of related disease markers. An extensive range of Biochip panels are available, each optimised to provide the best performance.

Cytokine Arrays
Cytokines play a vital role in the immune system and are known to be involved in the body’s response to a variety of inflammatory and infectious diseases. The over stimulation of these cytokines in response to infection is referred to as a ‘cytokine storm’ and strongly correlates with poor disease outcomes. Cytokine storms are a common complication of SARS-CoV-2 infection triggering viral sepsis, where viral replication and excessive, uncontrolled systemic inflammation may lead to pneumonitis, Acute Respiratory Distress Syndrome (ARDS), respiratory failure, shock, multiple organ failure, secondary bacterial pneumonia, and potentially death. Cytokines such as IL-6 appear to be elevated in patients with more severe illness and may be used to monitor response to treatment as levels decrease as the condition improves.
Randox offer a range of Quality control and External Quality Assessment (EQA) solutions designed to ensure the accuracy and reliability of patient testing.

Haematology
Decreased blood counts have been frequently identified in COVID-19 patients and may be indicative of a decreased immunological response to the virus, bacterial infection or consumption.

Blood Gas
Blood Gas tests are used to help diagnose and monitor critically ill patients in intensive care units (ICU) and emergency departments. Blood Gas testing continues to play a critical role in the management of COVID-19 patients providing an indication of a patient’s oxygenation levels. Monitoring of these levels will enable healthcare professionals to make ventilator adjustments if required.

Cardiac Markers
Increased levels of cardiac markers including Troponin and CK-MB may be associated with a higher mortality rate in COVID-19 patients.

Coagulation
Patients with severe COVID-19 infection are more likely to have coagulopathy, a condition in which the bloods ability to form clots is impaired. Activation of blood coagulation factors such as Prothrombin Time (PT) and D-dimer are significant predictors of disease severity and mortality.

Procalcitonin
Procalcitonin is an acute phase reactant released by most tissues during inflammation and is particularly associated with bacterial infection. Although it is not usually elevated in viral infections such as SARS-CoV-2 levels may increase as a result of severe systemic inflammation. It may also be used to help detect secondary infections.