Solutions
keyboard_arrow_downServices
keyboard_arrow_downSupport & Resources
keyboard_arrow_downCompany
keyboard_arrow_downContact
Features & Benefits of the Randox Bile Acids reagents
Excellent linearity
The Randox Bile Acids method is linear up to a concentration of 150 µmol/l
Exceptional correlation with standard methods
The Randox methodology was compared against other commercially available methods and the Randox Bile Acids assay showed a correlation coefficient of 0.99
Flexibility
Liquid and lyophilised reagents available for greater customer choice
Analyser protocols
Protocols are available for a range of analysers
Excellent stability
Stable to expiry when stored at +2 to +8°C
Ordering information
| Cat No | Size | |
|---|---|---|
| BI3863 (5th) | R1 2 x 18ml (L) R2 2 x 8ml | Enquire Kit Inserts RequestsView MSDSBuy Online |
| BI7982 (5th) | R1 6 x 50ml R2 6 x 18ml | Enquire Kit Inserts RequestsView MSDSBuy Online |
(L) Indicates liquid option
Instrument Specific Applications (ISA's) are available for a wide range of biochemistry analysers. Contact us to enquire about your specific analyser.
What are Bile Acids used for?
Clinical Significance
Liver Function
Measuring total bile acid (TBA) levels may prove useful for the detection of liver diseases such as viral hepatitis, mild liver injury through drug use and for further evaluation of patients with chronic hepatitis who were previously treated successfully. TBA levels may rise up to 100 times the normal concentration in patients with liver disease due to impairment of hepatic synthesis and extraction of bile acids. Measurement of TBA in serum can be used in the diagnosis and prognosis of liver diseases and may detect some forms of liver disease earlier than standard liver markers due to the correlation of TBA with liver function, rather than liver damage.
Bile Acid Deficiency
TBA deficiency is caused by a genetic error in one of the 17 enzymes that produce bile acids. Deficiency can lead to liver failure and even death in infants, therefore early detection is vital. People with TBA deficiency may exhibit symptoms, including:
- Vitamin deficiencies, specifically of fat-soluble vitamins such as A, D, E, and K
- Jaundice
- Stunted or abnormal growth
- Diarrhoea
- Loss of liver function
- Liver failure
Intrahepatic Cholestasis of Pregnancy
Intrahepatic cholestasis of pregnancy (ICP) or obstetric cholestasis is a pregnancy-specific liver disorder. It can be indicated by pruritus, jaundice, elevated TBA levels and/or serum transaminases and usually affects women during the second and third trimester of pregnancy. ICP is a condition that restricts the flow of bile through the gallbladder resulting in a build-up of TBA in the liver. Due to the build-up, Bile Acids leak into the bloodstream where they are detected at concerning levels. It is an extremely serious complication of pregnancy that can lead to the increased risk of premature birth or even stillbirth as such it is vital that women with the condition are monitored carefully.
According to several reports TBA levels in ICP can reach as high as 100 times the upper limit of a normal pregnancy. It has been reported that a doubling in maternal serum TBA levels, results in a 200% increased risk of stillbirth. Additionally, bile acids can affect the foetal cardiovascular system as it has been found that there are often cardiac rhythm disturbances in the foetus due to the elevated TBA in circulation.
There are several risk factors associated with ICP such as family history, use of oral contraceptives, assisted reproduction techniques and multiple gestation. Genetic influence accounts for approximately 15% of ICP cases. Dietary selenium is a contributing environmental factor as serum selenium levels often decrease throughout pregnancy. Further to this, incidences of ICP rise in the winter months and are most likely due to the fact selenium levels are naturally less during these months. In healthy pregnancies, there is very little increase in TBA levels although a slight increase is likely to be seen in the third trimester.
Measurement of TBA in serum is thought to be the most suitable method of diagnosing and monitoring ICP.
Biological Significance
Bile acids are water-soluble and amphipathic end products of cholesterol metabolism formed in the liver. Bile is stored in the gall bladder and released into the intestine when food is consumed. The fundamental role of bile acids is to aid in the digestion and absorption of fats and fat-soluble vitamins in the small intestine. In doing so, bile acids have five physiological functions within the body as shown below:

Inadequacies of traditional bile acid assays
Determining the cause and extent of liver damage is important in guiding treatment decisions and preventing disease progression. Standard liver function tests include; ALT, AST, ALP, GGT and Bilirubin. The measurement of TBA is most beneficial in conjunction with these standard liver tests and offers unrivalled sensitivity allowing identification of early stage liver dysfunction.
There are several commercial methods available for the detection and measurement of TBA in serum. Traditional TBA tests based on the enzymatic method use nitrotetrazolium blue (NBT) to form a formazan dye. The reaction is measured at 546nm and the intensity of the colour is proportional to the concentration of bile acids.
Newer methods such as the enzyme cycling method or fifth generation methods offer many advantages including greater sensitivity, liquid reagents, small sample volumes and reduced instrument contamination from formazan dye. Additionally, the fifth generation assay does not suffer from interference from lipaemic or haemolytic samples. Both lipaemia and haemolysis are common in new-borns and pregnant women.
Enzyme cycling methods offer superior analytical performance, two reactions are combined. In the first reaction, bile acids are oxidised by 3-α hydroxysteroid dehydrogenase with the subsequent reduction of Thio-NAD to Thio-NADH. In the second reaction, the oxidised bile acids are reduced by the same enzyme with the subsequent oxidation of NADH to NAD. The rate of formation of Thio-NADH is determined by measuring the specific absorbance change at 405nm. Enzyme cycling means multiple Thio-NAD molecules are generated from each bile acid molecule giving rise to a much larger absorbance change and signal amplification, increasing the sensitivity of the assay.
The assay principle is demonstrated in the diagram below:
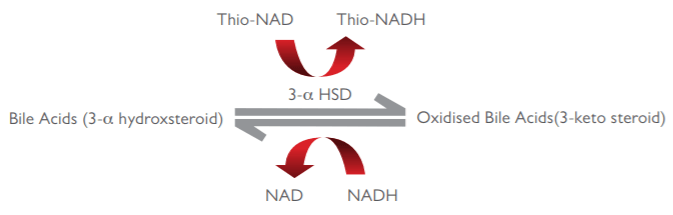
The Randox fifth generation assay utilises the advanced enzyme cycling method which displays outstanding sensitivity and precision compared to traditional enzymatic based tests. The assay shows excellent linearity of up to 188 µmol/l with the normal upper range of TBA in a fasting serum sample being at 10 µmol/l. The liquid ready-to-use reagent is available along with complementary controls and calibrators for a complete testing package.
Get in touch to discover more
To find out more about Bile Acids Reagent and other diagnostic reagents, enquire now.