Alzheimer’s Disease: The Role of Apolipoprotein E
Alzheimer’s Disease: The Role of Apolipoprotein E

Alzheimer’s Disease: The Role of Apolipoprotein E
Raising awareness of Alzheimer’s Disease
Every year we celebrate Alzheimer’s Day on 21st September to help raise awareness around dementia. Dementia is the medical name attributed to a set of symptoms affecting the brain, including: difficulties with problem solving, thinking, language and memory loss. AD is the most common form of dementia accounting for 60 – 80% of cases and it is believed that half of patients with Alzheimer’s dementia (dementia due to AD) have Alzheimer’s disease 1, 2.
About Alzheimer’s Disease (AD)
AD is one of the most devastating and complex diseases characterised by:
- Neurodegeneration resulting in memory loss 2
- Neurofibrillary tangles composed of tau amyloid fibrils which associates with synapse loss 2
- Accumulation of β-amyloid (Aβ) plaques 2
- Other cognitive functions 2
Figure 1: Alzheimer’s Disease Demographic, 2019 3

It is believed that AD is expected to begin 20 years prior to symptom onset, as the small changes in the functioning of the brain are unnoticeable to the person affected. Overtime, the symptoms progress and begin to interfere with the patient’s ability to perform everyday tasks. The final stages of AD leaves the patient bed-bound, requiring 24/7 care. Ultimately, AD is fatal. Age has been identified as a risk factor for AD with 10% of people over the age of 65 affected. Moreover, AD has been recognised as a leading cause of morbidity and the sixth leading cause of mortality, but the fifth leading cause of death in over 65’s in the US .3
Physiological Significance of Apolipoprotein E
Apolipoprotein E (Apo E) is a lipoprotein composed of 299 amino acids with a molecular weight of 34kDa. Apo E is responsible for the regulation of homeostasis through the mediation of lipid transport from and to bodily cells and tissues. Apo E comprises of three common isoforms: apo E2, apo E3 and apo E4. The apo E isoforms differ due to differences in either the 112 and 158 amino acids, whether either arginine (ARG) or cysteine (CYS) is present 4.
Apo E3 is the parent form of apo E and is responsible for the clearance of triglyceride-rich lipoproteins. Apo E3 is associated with normal lipid plasma concentrations. Apo E2 is the rarest of the apo E isoforms and differs slightly compared to the apo E3 isoform through the substitution of a single amino acid, ARG158Cys, located near the low-density lipoprotein receptor (LDLR) recognition site.
Apo E2 displays impaired binding to the receptor, prohibiting the clearance of triglyceride-rich lipoprotein remnant particles. Apo E2 is strongly associated with type-III hyperlipoproteinemia. Apo E3 also differs from apo E4, again through the substitution of a single amino acid, Cys112Arg. The main difference between apo E3 and apo E4 is that apo E4 is unaffected by the binding of the isoform to LDLR. However, apo E4 is strongly associated with dyslipidemia 5. Fig. 2 provides a visual representation of the variations in the Apo E isoforms.
Figure 2: Variations in the Apo E Isoforms 4

Apo E is expressed in numerous bodily organs with the liver presenting with the highest expression followed by the brain. Astrocytes and, to a lesser extent, microglia are the major cells responsible for the expression of apo E in the brain. In the brain, apo E, apo J and apo A-1 are predominantly expressed on distinct high-density-like lipoprotein particles. Whilst apo A-1 is the major apolipoprotein of high-density lipoproteins (HDL), in the central nervous system (CNS), apo E is the predominant apolipoprotein of HDL-like lipoproteins. HDL-like lipoproteins are the only lipoproteins present in the CNS. It is believed that the cholesterol released from apo E supports synaptogenesis 6.
Clinical Significance of Apolipoprotein E in Alzheimer’s Disease
Whilst apo E3 is the most abundant of the three isoforms, apo E4 has been known for decades to be the most significant genetic risk factor for late-onset AD. Inheriting the one copy of the apo E4 gene increases the risk of AD 2-3-fold, whilst inheriting two copies increases the risk of AD up to 12-fold 7. Whilst the underlying mechanism of apo E’s contribution to AD risk is still unclear and debatable, apo E has been identified as promoting amyloid β (Aβ) deposition and clearance as well as neurofibrillary tangles in the brain. Interestingly, Aβ-independent pathways exist for apo E in AD, which led to the unearthing of the new roles of apo E including the most recent, iron metabolism and mitochondria dysfunction 8, 9. Captivatingly, sex-related hormones may play a role in AD in apo E4 carriers as AD has been recognised to be more pronounced in women 10. Apo E4 has also been identified as impairing lipid transport, microglial responsiveness, glucose metabolism, synaptic plasticity and integrity, and cerebrovascular function and integrity. Some of these pathogeneses are independent of Aβ pathways. Furthermore, therapeutic strategies are aiming to modulate the quantity, lipidation, structural properties, Aβ interaction and receptor expression of Apo E 11.
Key Features of the Randox Apolipoprotein E Assay
Randox are one of the only manufacturers to offer the apo E assay in an automated clinical chemistry format. Utilising the immunoturbidimetric method, the Randox apo E assay is available in a liquid ready-to-use format. Not only does the Randox apo E suffer from limited interferences from bilirubin, haemoglobin, intralipid® and triglycerides for truly accurate results, it has an excellent measuring range of 1.04 – 12.3mg/dl for the comfortable detection of clinically important results. Moreover, apolipoprotein calibrator and controls are available for a complete testing package. Applications are available detailing instrument-specific settings for the convenient use of the Randox apo E assay on a wide range of clinical chemistry analysers.
Biochip Technology – Alzheimer’s Array
Utilising the Biochip Technology, Randox have developed an array to identify the risk of Alzheimer’s disease in just 3 hours with one effective test. In addition to a rapid and accurate diagnosis, this also introduces both cost and time-saving benefits. The apo E4 array is a research use only product developed for the Evidence Investigator, a semi-automated benchtop immunoassay analyser which can process up to 2376 test per hour as well as up to 44 analytes screened per biochip. The apo E4 array measures both total apo E protein levels and apo E4 protein levels directly from plasma samples as well as using a ratio, it can classify patients as negative or positive for apo E4. In turn, we can then assess their risk for the development of Alzheimer’s disease.
Related Products
Randox Reagents Homepage
Evidence Series Immunoanalysers
RX Series Analysers
References
[1] Alzheimer’s Society. Alzheimer’s disease. [Online] [Cited: September 2, 2019.] https://www.alzheimers.org.uk/about-dementia/types-dementia/alzheimers-disease.
[2] Gaugler, Joseph, et al. 2019 Alzheimer’s Disease Facts and Figures. s.l. : Alzheimer’s Association, 2019.
[3] 2014 Update of the Alzheimer’s Disease Neuroimaging Initiative: A review of papers published since its inception. Weiner, Michael W, et al. 6, San Francisco : Alzheimer’s & Dementia: The Journal of the Alzheimer’s Association, 2015, Vol. 11.
[4] Apolipoprotein E and Alzheimer disease: risk, mechanisms, and therapy. Liu, Chia-Chen, et al. 2, Fujian : Nature Reviews Neurology, 2013, Vol. 9.
[5] Apolipoprotein E isoforms and lipoprotein metabolism. Phillips, Michael C. 9, Philadelphia : IUBMB Journals, 2014, Vol. 66.
[6] The Role of Apolipoprotein E in Alzheimer’s Disease. Kim, Jungsu, Basak, Jacob M and Holtzman, David M. 3, St Louis : Neuron, 2009, Vol. 63.
[7] Dacks, Penny. What ApoE Means For Your Health. Cognitive Vitality. [Online] November 16, 2016. [Cited: September 11, 2019.] https://www.alzdiscovery.org/cognitive-vitality/blog/what-apoe-means-for-your-health.
[8] The Complex Role of Apolipoprotein E in Alzheimer’s Disease: an Overview and Update. Mahoney-Sanchez, Laura, et al. 3, Parkville : Journal of Molecular Neuroscience, 2016, Vol. 60.
[9] Understanding the Role of ApoE Fragments in Alzheimer’s Disease. Muñoz, SS, Gerner, B and Ooi, L. 6, Wollongong : Neurochemical Research, 2019, Vol. 44.
[10] ApoE4: an emerging therapeutic target for Alzheimer’s disease. Affieh, Mirna, Korczyn, Amos D and Michaelson, Daniel M. 64, s.l. : BMC Medicine, 2019, Vol. 17.
[11] Apolipoprotein E and Alzheimer disease: pathobiology and targeting strategies. Yamazaki, Yu, et al. 9AB, s.l. : Nature Reviews Neurology, 2019, Vol. 15.
COVID-19 Management of Kidney Injured Patients – CKD & AKI
COVID-19 Management of Kidney Injured Patients
Analysis of COVID-19 patients revealed that Acute Kidney Injury (AKI) is common and associated with a very high mortality rate highlighting the need for more accurate patient testing. Further to this the National Institute for Health and Care Excellence (NICE) recommend that all COVID-19 patients are assessed for AKI on admission to hospital and their condition monitored throughout their stay. The complications with serum creatinine measurement alone for the detection of impaired kidney function are well known. To address this issue, Randox have developed three multi marker kidney function arrays for early detection of renal impairment. Individuals with pre-existing kidney injury are at an increased risk of COVID-19, those with severe CKD (stages 3-5) are at a higher risk of complications.
Utilising patented Biochip Technology, the Randox Chronic Kidney Disease (CKD) and Acute Kidney Injury (AKI) arrays could improve COVID-19 risk stratification whilst monitoring the effectiveness of treatment.


Randox Chronic Kidney Disease (CKD) Array I (7-plex)
EGF regulates renal cell proliferation, fibrosis and inflammation and is produced in response to renal injury.
IL-8 endothelial-derived chemokine involved in recruiting neutrophils to sites of injury and stimulating their response.
sTNFR1 is used to identify an increase in inflammatory conditions such as CKD.
FABP1 binds long-chain fatty acids, contributing to reducing oxidative stress in the kidneys.
sTNFR2 is used to identify an increase in inflammatory conditions such as CKD.
D-Dimer is a fibrin degradation product, and an index of both coagulation and fibrinolysis.
MIP-1 alpha plays a roles in inflammatory responses at sites of injury or infection.
Randox Chronic Kidney Disease (CKD) Array II (4-plex)
CRP is an acute phase reactant involved in inflammation.
Cystatin C is well recognised marker of kidney filtration dysfunction and injury.
C3a des Arg is a representative of complement component C3a which produces local inflammatory responses.
NGAL is among the current state-of-the-art in CKD biomarkers.
Randox Acute Kidney Injury (AKI) Array (4-plex)
This marker is highly upregulated in kidney tubule cells following nephrotoxic injury severe enough to result in acute renal failure, acute tubular necrosis or acute tubulo-interstitial nephropathy.
Due to its small size and basic pH, Cystatin C is freely filtered by the glomerulus. It is then reabsorbed by tubular epithelial cells and subsequently metabolized. Accumulation of Cystatin C in urine is specific for tubular kidney damage and suggests reduced reabsorption at the proximal tubules as a result of toxicant-induced kidney injury.
Expression of Clusterin is upregulated following a variety of renal injuries and is detectable in urine following acute kidney injury induced by administration of nephrotoxic agents. This occurs before the profound renal transformations that give rise to changes in creatinine and BUN.
KIM-1 is a 30kDa type 1 transmembrane glycoprotein found on actvated CD4+ T cells. It is undetectable in healthy kidney tissue but is expressed at very high levels in proximal tubule epithelial cells in the kidney after toxic injury.
The Evidence Investigator
Meet the Evidence Investigator
The Randox CKD & AKI arrays have both been developed for the Evidence Investigator, a semi-automated benchtop immunoassay analyser.
The CKD & AKI array’s would improve COVID-19 risk stratification whilst monitoring the effectiveness of treatments by simultaneously and quantitatively detecting multiple serum biomarkers of kidney damage-related analytes from a single sample.



Want to know more?
Contact us or visit our Investigator Webpage
Alzheimer’s Disease Array
Rapid Identification of Alzheimer's Disease Risk
The Randox ApoE4 Array is a rapid and highly sensitive blood test facilitating direct ApoE4 genotyping without the need for molecular genotyping. ApoE exists as three common isoforms; ApoE2, ApoE3 and ApoE4. As such, six common ApoE genotypes exist in the general population. Alzheimer’s Disease risk is significantly increased in individuals with the ApoE4 allele. The below table provides an indication of risk associated with the six common ApoE genotypes.




Biomarkers Tested
ApoE is a major cholesterol carrier, responsible for lipid homeostasis by mediating lipid transport from one tissue or cell type to another. In the central nervous system, ApoE is mainly produced by astrocytes, and transports cholesterol to neurons via ApoE receptors. ApoE exists as three common isoforms; ApoE2, ApoE3 and ApoE4.
ApoE4 is established as the strongest genetic risk factor for Alzheimer’s Disease. ApoE4 triggers inflammatory cascades that cause neurovascular dysfunction, including blood-brain barrier breakdown, leakage of blood-derived toxic proteins into the brain and reduction in the length of small vessels.
Apo E4 is one of three common isoforms of Apo E and is recognised as a major genetic risk factor the development of Alzheimer’s disease. Apo E4 triggers inflammatory cascades that cause neurovascular dysfunction, including blood-brain barrier breakdown, leakage of blood-derived toxic proteins into the brain and reduction in the length of small vessels.
The Evidence Investigator
Meet the Evidence Investigator
The Randox ApoE4 Array (EV4113) is a research use-only product developed for the Evidence Investigator, a semi-automated benchtop immunoassay analyser.
The ApoE4 Array (EV4113) simultaneously measures both total ApoE and ApoE4 protein levels directly from a plasma sample. The Apo E4/total ApoE ratio can classify the ApoE4 status of the plasma sample as negative or positive. Furthermore, the Array has demonstrated potential to classify ApoE4 positive plasma samples as being derived from a heterozygous (one copy of ApoE4 gene) or a homozygous (two copies of ApoE4 gene) individual. Therefore, risk for the development of Alzheimer’s Disease can be assessed.



Publications
Want to know more?
Contact us or visit our Cerebral Array webpage.
CRP Assay
Reagent | C-Reactive Protein (CRP)
C-Reactive Protein (CRP): The Most Sensitive Acute Phase Reactant
Benefits of the Randox Assay


Limited Interference
The Randox CRP assay has shown to have limited interference from conjugate and free bilirubin, haemoglobin, Intralipid® and triglycerides.


Liquid ready-to-use
The Randox CRP assay is available in a liquid ready-to-use format for convenience and ease-of-use.


Stable to expiry
The Randox CRP assay is stable to expiry when stored at +2oC to +8oC.


Applications available
Applications available detailing instrument-specific settings for the convenient use of the Randox CRP assay on a variety of clinical chemistry analysers.


Dedicated calibrator and controls available
Dedicated CRP calibrator and controls available offering a complete testing package.
Ordering Information
| Cat No | Size | ||||
|---|---|---|---|---|---|
| CP7950 | R1 7 x 20ml (L) R2 2 x 12ml | Enquire | Kit Insert Request | MSDS | Buy Online |
| CP9742 | R1 6 x 66ml (L) R2 6 x 13ml | Enquire | Kit Insert Request | MSDS | Buy Online |
| CP3826 | R1 6 x 20ml (L) R2 3 x 9ml | Enquire | Kit Insert Request | MSDS | Buy Online |
| (L) Indicates liquid option | |||||
Instrument Specific Applications (ISA’s) are available for a wide range of biochemistry analysers. Contact us to enquire about your specific analyser.
More Information
C-reactive protein (CRP) is the dominant acute phase protein in mammals 1. Elevated levels of CRP are not disease specific but are synthesised, by hepatocytes, in response to pro-inflammatory cytokines during inflammatory and/or infectious processes 2, 3. The physiological function of CRP is the enhancement of cell-mediated immunity through the promotion of phagocytosis, accelerated chemotaxis and the activation of platelets 1.
Known as a biomarker of acute inflammation, many large-scale prospective studies demonstrate the association between CRP and chronic inflammation, including: cardiovascular disease (CVD), cerebrovascular accident (CVA) (ischaemic stroke), Alzheimer’s Disease, and age-related macular degeneration 2.
CRP is a major cardiovascular disease (CVD) risk factor. A CRP level of >10mg/l correlates with a >4% risk of developing a fatal CVD event in 10 years, including: myocardial infarction, peripheral arterial disease, stroke and sudden cardiac death. Inflammation is an integral part in the development and rapid progression of coronary heart disease (CHD) 4.
The coronary artery disease (CAD) process is characterised by increasing levels of inflammatory biomarkers. CRP is not only an excellent biomarker or mediator of atherosclerosis but is a strong independent marker in the prediction of adverse CV events, including: myocardial infarction, ischaemic stroke, and sudden cardiac death. CPR can be utilised as a clinical guide for the diagnosis, management and prognosis of CVD 5.
The development of a cerebrovascular accident (CVA) (stroke) is the result of longstanding vascular inflammation, thrombosis, plaque rupture and subsequent brain ischaemia or infarction. Elevated CRP levels is associated with the development of CVA 6.
High sensitivity CRP (hsCRP) plays an important role in the early diagnosis, prognosis, long and short death risk and prediction of etiological subtypes of stroke 7. Find out more about the Randox hsCRP assay.
Alzheimer’s disease (AD) is the most common form of dementia, accounting for 60-80% of dementia cases 7. CRP is believed to be involved in the pathophysiology of cerebral small vessel disease (CSVD) and neurodegeneration 8. CRP levels are associated with cognitive impairment 9. Inflammation should be considered as a target treatment, with the aim of delaying the progression of subclinical brain damage and cognitive decline 8. Midlife elevations in CRP are associated with an increased risk of developing AD. Patients with AD, CRP elevations continues to predict increased dementia severity suggestive of a possible pro-inflammatory endophenotype in AD 10.
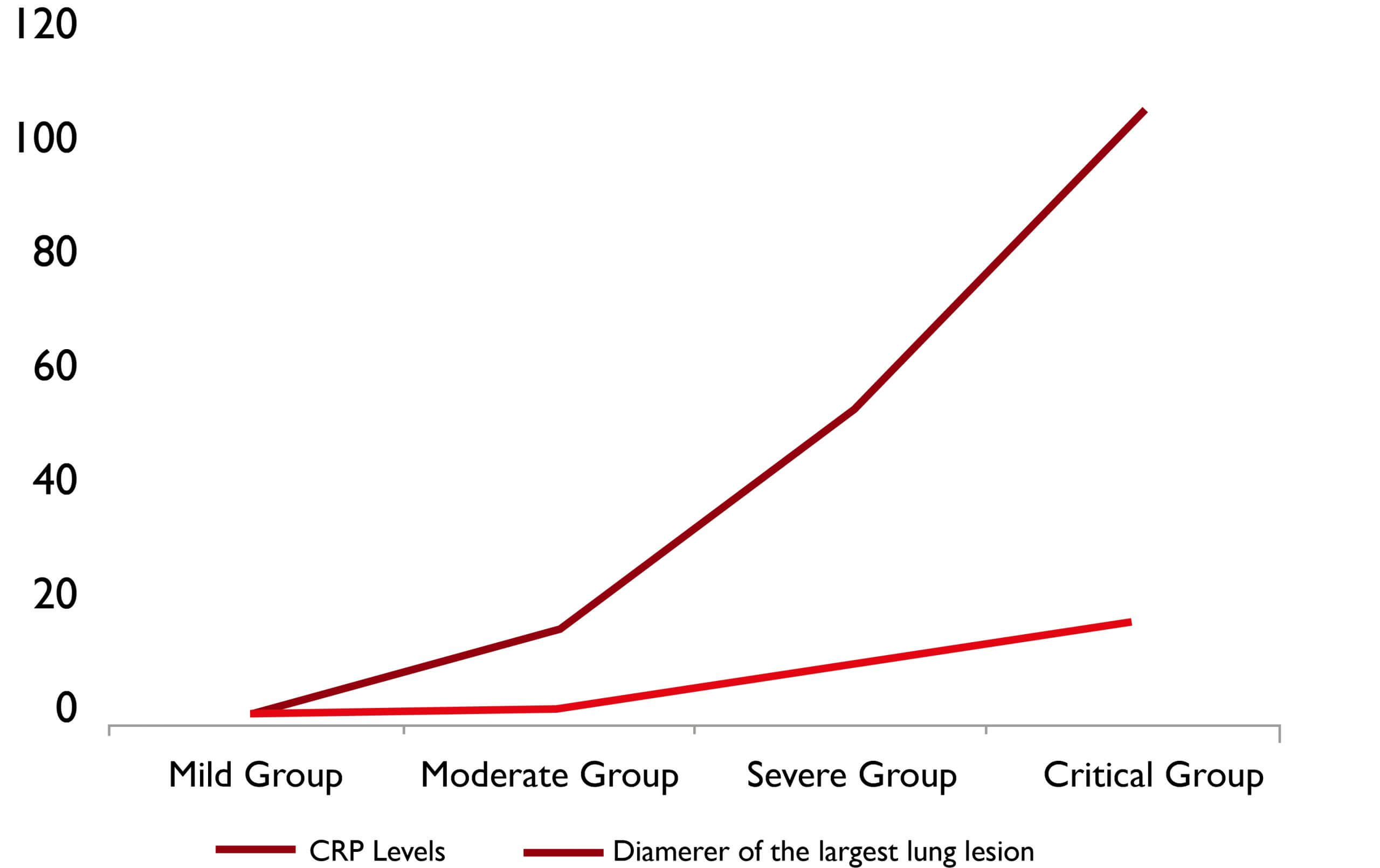
In COVID-19 patients, CRP testing has proved to perform well in discriminating disease severity and predicting adverse outcomes 11. CRP levels positively correlates with lung lesions, reflecting disease severity, and should be considered a key indicator in disease monitoring (see fig. 1) 12.
CRP levels are associated with computed tomography (CT) scores and COVID-19 disease development, with CRP levels increasing in the initial stage of severe COVID-19, prior to the CT findings 13.
Elevated CRP levels have been identified in 86% of patients admitted to hospital. CRP measurements are useful in diagnosis, assessing prognosis and monitoring for clinical improvements or deterioration 14.
Fig. 1. Relation between CRP levels, clinical severity, and lung lesions 12

CRP Calibrator
CRP Control
Specific Protein EQA
Reagents Homepage
References
How important is Homocysteine research for Alzheimer’s disease?
It is widely recognised that high homocysteine levels in the blood (hyperhomocysteinemia) can cause inflammation in the blood vessels, which in turn may lead to atherogenesis and ischemic injury. High homocysteine levels are therefore a possible risk factor for coronary artery disease (CAD), or heart disease.
However a new study published in the Journal of Thrombosis and Haemostasis in April 2016 (conducted by Young Cheul Chung and colleagues, from Rockefeller University in New York City) has looked into the growing evidence to suggest that hyperhomocysteinemia is also correlated with Alzheimer’s disease and vascular dementia. This study was undertaken to clarify the specific role of elevated homocysteine levels in Alzheimer’s disease pathophysiology.
The study was carried out on mice, and showed a promising link between high levels of homocysteine and Alzheimer’s disease. It showed that diet-induced hyperhomocysteinemia in an Alzheimer’s disease mouse model leads to severe cerebral amyloid angiopathy and parenchymal amyloid-β deposition, as well as significant impairments in learning and memory, suggesting that elevated levels of plasma homocysteine and its metabolite, homocysteine thiolactone, contribute to Alzheimer’s disease pathology.
Commenting on an earlier study, Dr Susanne Sorensen, Head of Research at Alzheimer’s Society UK said that the molecule [homocysteine] is carried by everyone, but those who go on to develop some dementias appear to have higher levels of the compound. She also stressed that research is needed to establish just what role if any homocysteine plays in the development of dementia and how best to keep levels of the molecule low.
Alzheimer’s disease is a progressive mental decline that can occur in middle to old age, due to a degeneration of the brain. It is the most common cause of premature senility, and is also the most common form of dementia, affecting 62% of those diagnosed. Vascular dementia is another form, affecting 17% of those diagnosed.
Paul McGivern, Clinical Chemistry R&D Manager at global healthcare company Randox Laboratories, has commented
“Dementia is a terminal condition and with 850,000 people living with dementia in the UK, there is an urgent need for further research into the link between Alzheimer’s disease and homocysteine levels. If we can better establish this link, it may give future researchers the tools necessary to find a prevention, or even a cure to this condition.”
With the number of dementia sufferers set to rise to over 1 million by 2025, soaring to 2 million by 2051, the need for further research into the link between homocysteine levels and Alzheimer’s disease has never been more pressing.
For health professionals
Randox Laboratories offer an automated test for the biochemistry measurement of homocysteine. This is available for use on a wide range of manufacturer’s analysers. Please contact reagents@randox.com for further information, or to request a kit insert or application.











