RIQAS Point of Care EQA
RIQAS Point of Care EQA
Designed to improve the quality of Point of Care Testing (POCT) in locations such as pharmacies, GP surgeries, hospital out patient departments, sports clinics, supermarkets, diagnostic/treatment and walk-in centres.
RIQAS Point of Care EQA provides independent evidence of the accuracy and reliability of test results.
Why RIQAS Point of Care?
Programme Offering
Tests
Role
Matrix
Lipids (Total Cholesterol & HDL Cholesterol)*
- Risk Factor for heart Disease
- Monitoring lipid lowering therapy
Whole Blood
HbA1c (Glycated Hemoglobin)*
- Diagnosing diabetes mellitus
- Monitoring treatment
- Encouraging self-management
Whole Blood
CRP (C Reactive Protein)*
- Early detection of infectious disease
- Identifying need for antibiotic treatment
Whole Blood
Glucose / Ketones
- Diagnose and monitor diabetes
- Monitor the presence of hypoglycemia (low blood glucose) and hyperglycemia (high blood glucose)
- To determine whether excessive ketones are present in the blood, to detect diabetic ketoacidosis (DKA)
Serum
International Normalised Ratio (INR)
- Used to measure the effect of anticoagulant drugs such as warfarin
- Help diagnose a bleeding disorder; to help estimate the severity of liver disease
Whole Blood
Key Cycle Dates
RQ9181 Distribution Month Sample Distributed Result Submission Deadline January 2024 8th January 17th January February 2024 5th February 14th February March 2024 4th March 13th March April 2024 2nd April 10th April May 2024 6th May 15th May June 2024 3rd June 12th June July 2024 1st July 10th July August 2024 5th August 14th August September 2024 2nd September 11th September October 2024 7th October 16th October November 2024 4th November 13th November December 2024 2nd December 11th December What Participants Say
What participants say
Our unrivalled commitment to quality and service ensures high levels of customer satisfaction, this is evident from the responses to our latest customer satisfaction survey:
“All in all a quick and efficient service”
“Good online system”
“Very helpful team”
“Excellent training”
“They are an experienced team”
“Very satisfied with the service that we receive”
“Very good value for money”
“The website is great”
The Importance of Quality Assurance
Importance of quality assurance
Quality assurance is an essential aspect of any clinical/diagnostic testing service and is aimed at ensuring the accuracy and reliability of patients’ results. The right result allows the right clinical advice to be offered in a timely manner. Quality assurance operates at two levels:Internal Quality Control
Internal Quality Control includes operator training/ competency assessment, analyser/ test system maintenance, and adherence to policies/ processes. Whilst some point of care analysers include inbuilt quality checks, cross-check analysis against samples with known levels provides immediate assurance and evidence that a patient’s result is safe to report.External Quality Assessment
External Quality Assessment involves analysis of samples with unknown levels that have been distributed by an external organisation. Participants are informed how their results compare with other participants, hence providing independent evidence of performance. Increasingly, participation in an external quality assessment scheme is becoming a mandatory requirement where health and healthcare services are being provided.
EQA provides assurance to both staff and customers that testing provides accurate and reliable results.
Want to know more?
Contact us or download the RIQAS Point of Care catalogue to learn more.
Continue Reading
Frequently Asked Questions
RIQAS
Contact Us
RIQAS Point of Care EQA
Designed to improve the quality of Point of Care Testing (POCT) in locations such as pharmacies, GP surgeries, hospital out patient departments, sports clinics, supermarkets, diagnostic/treatment and walk-in centres, RIQAS Point of Care EQA provides independent evidence of the accuracy and reliability of test results. Randox International Quality Assessment Scheme (RIQAS) is the world’s largest EQA scheme with over 55,000 participants in more than 134 countries.
Why RIQAS Point of Care?
About RIQAS Point of Care
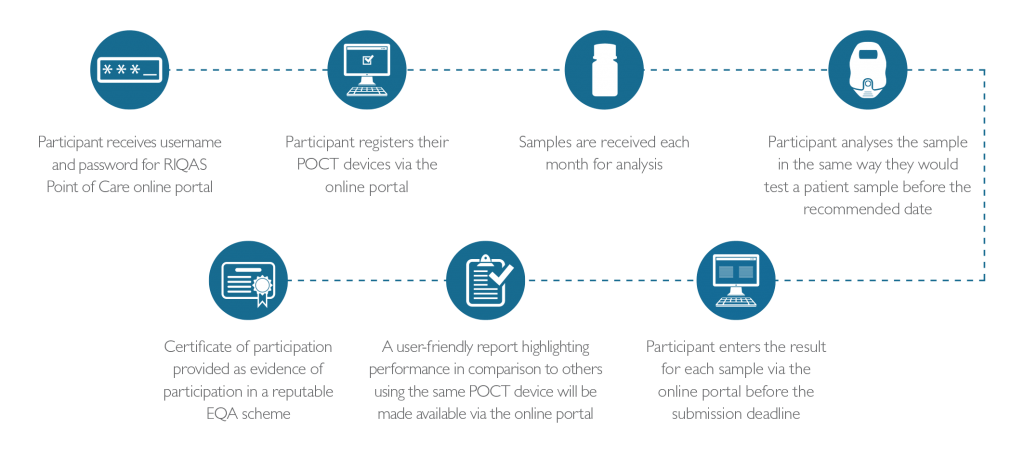
How it Works
Tests and Analysers
Test Role Matrix Lipids (Total Cholesterol & HDL Cholesterol) • Risk factors for heart disease
• Monitoring lipid lowering therapy
Whole Blood HbA1c (Glycated Haemoglobin) • Diagnosing diabetes mellitus
• Monitoring treatment
• Encouraging self-management
Whole Blood C-Reactive Protein (CRP) • Early detection of infectious disease
• Identifying need for antibiotic treatment
Whole Blood Glucose/Ketones • Diagnose and monitor diabetes
• Monitor for the presence of hypoglycaemia
(low blood glucose) and hyperglycaemia (high blood glucose)
• To determine whether excessive ketones are present in the blood, to detect diabetic ketoacidosis (DKA)
Serum International Normalised Ratio (INR) • Used to measure the effect of anticoagulant
drugs such as warfarin
• Help diagnose a bleeding disorder; to help
estimate the severity of liver disease
Plasma Note – The RIQAS Point of Care range is constantly expanding to include new tests and analysers. Please contact us if your desired analyser or test is not currently displayed.Ordering Information
Panel Catalogue Number Lipids RQ9181/A Lipids + 1 panel RQ9181/B Lipids + 2 panels RQ9181/C Additional Sample RQ9181/D Glucose and Ketones RQ9188 INR RQ9189 Pipette Tips RQ9182 Bulbous pipette RQ9183 What Participants Say
Our unrivalled commitment to quality and service ensures high levels of customer satisfaction, this is evident from the responses to our latest customer satisfaction survey:
“All in all a quick and efficient service”
“Good online system”
“Very helpful team”
“Excellent training”
“They are an experienced team”
“Very satisfied with the service that we receive”
“Very good value for money”
“The website is great”Key Cycle Dates
RQ9181 Distribution Month Sample Distributed Result Submission Deadline January 2024 8th January 17th January February 2024 5th February 14th February March 2024 4th March 13th March April 2024 2nd April 10th April May 2024 6th May 15th May June 2024 3rd June 12th June July 2024 1st July 10th July August 2024 5th August 14th August September 2024 2nd September 11th September October 2024 7th October 16th October November 2024 4th November 13th November December 2024 2nd December 11th December Importance of Quality Assurance
Quality assurance is an essential aspect of any clinical/diagnostic testing service and is aimed at ensuring the accuracy and reliability of patients’ results. The right result allows the right clinical advice to be offered in a timely manner. Quality assurance operates at two levels:Internal Quality Control
Internal Quality Control includes operator training/ competency assessment, analyser/ test system maintenance, and adherence to policies/ processes. Whilst some point of care analysers include inbuilt quality checks, cross-check analysis against samples with known levels provides immediate assurance and evidence that a patient’s result is safe to report.External Quality Assessment
External Quality Assessment involves analysis of samples with unknown levels that have been distributed by an external organisation. Participants are informed how their results compare with other participants, hence providing independent evidence of performance. Increasingly, participation in an external quality assessment scheme is becoming a mandatory requirement where health and healthcare services are being provided.
EQA provides assurance to both staff and customers that testing provides accurate and reliable results.Want to know more?Visit our Importance of EQA page to learn more.
Want to know more?
Contact us or download the RIQAS Point of Care catalogue to learn more.
Continue Reading
Frequently Asked Questions
RIQAS
Contact Us
Point of Care Testing (POCT) Explained


Point of Care Testing (POCT) is the delivery of a test at the point in time at which the result will be used to make a decision and taking appropriate action resulting in an improved health outcome. It is also known as near patient, bed-side, extra-laboratory, decentralised, and ancillary testing [1]. It has been shown to reduce hospital stay time, reduce complications, and improve adherence to treatment [2].
Point of care testing is not a recent practice; many early diagnostic tests were administered at the bedside. However, analytical technology has progressed and multiple tests can be performed within minutes in a laboratory. Recently, this technology has been put into the hands of the staff near the patients [2]. There are two types of technology, benchtop analysers and hand held devices. Bench top systems are just smaller versions of laboratory analysers but some steps are automated. Hand held devices are simple in appearance but complex internally, they can manage several tasks including, adding reagents, separating cells from plasma, and reading colour or other measures.
Results can be obtained faster, allowing for more immediate decisions meaning treatment can begin sooner. Patients can live a longer and higher quality life, helped by a reduction in the length of hospital stays.
Some benefits of POCT [2]:
Key objective
The main objective of Point of Care Testing is to generate results more quickly so that appropriate treatment can be provided, resulting in an improved patient outcome.
Management
Accurate and reliable results can only be obtained if the patient and sample are treated correctly. Point of care testing is likely to be performed by staff with a limited technical background, so training and quality control are vital.
Outcomes
Proper analysis technique alone is not enough to ensure an accurate decision; any test will only be beneficial if the appropriate action is taken based on the result. The effectiveness of POCT is assessed in terms of the overall outcome of the patient.
There are three phases in the POCT cycle: pre-analytical phase, analytical phase, and post-analytical phase. About 90% of quality issues are attributed to the pre-analytical and post-analytical phases [3]. These errors are mainly attributed to user error and can be caused by a number of issues including, selecting the wrong POCT device, not following manufacturer instructions, inadequate training, not adhering to appropriate QC practices, and many more.
The errors can usually be mitigated by implementing an action plan and ensuring it is executed exactly as designed, deviation from the action plan will lead to errors. Errors in POCT diagnostics can lead to misdiagnosis, improper treatment, costly follow-up procedures, and death.
Some strategies for improvement:
Internal Quality Control and External Quality Assessment is conducted to monitor the stability of the analytical measurement system and to alert the operator to a change that may lead to a medically significant error [6].
A study by Price, Smith and Bruel [8] was conducted on a number of labs over a period of time of up to 15 years. They discovered that test result performance improved with time and was associated with regular participation in External Quality Assessment (EQA) schemes and with the use of internal quality control (IQC) procedures.
Internal Quality Control
Internal Quality Control (IQC) is used to assess the day-to-day consistency of assay performance, providing quality assurance for patient results. IQC activities are among the ten most common POCT deficiencies. These may include performing and documenting quality control testing and taking the correct action for outliers [4]. This poor performance could be attributed to how IQC is viewed in POCT; users may lack appreciation of the potential for errors and may see the analyser as infallible, they likely see IQC as an additional workload as opposed to part of their testing routine.
CLSI regulations require risk assessment for each stage of patient testing alongside an implementation of a quality control plan. Below are some suggestions for how IQC should be conducted for POCT.
Conduct
IQC should be conducted when: a new lot of consumable is used; a patient result is queried; after maintenance; the device has been physically insulted. IQC should be conducted by the usual device operator so assurance can be provided for the whole testing process.
Training
ISO 22870 requires POCT users should be trained in the theory and practice of IQC [5]. Staff should be trained in every aspect of POCT including storage, preparation, frequency, documentation and basic troubleshooting.
Material
QC material for POCT should be obtained from a third party provider and not rely on material provided by the device manufacturer, the benefits of which are well documented. It should also contain analytes at clinically relevant concentrations, be provided ready-to-use, and be stable at ambient temperatures.
Results
All IQC results must be recorded with the date, time, user, decision to accept or reject, and any actions taken as appropriate. Locally assigned ranges alongside analyte-specific rules should be used to maximise error detection. An example of how IQC could be recorded and an action flowchart can be seen in Fig. A below.
Troubleshooting
There should be a protocol for required actions following a failed IQC. Any troubleshooting should be developed with knowledge of the most common errors and user capability.
Review
A monthly review should be conducted to identify persistent failures and trends.
The cost of IQC may also be a factor in resistance to IQC, however, while it is difficult to quantify, the cost of not conducting it may be greater in terms of human harm. A whitepaper is available detailing IQC in POCT (download).
Fig. A Examples of a manual IQC documentation, adapted from the Australian Government’s POCT General Practice [4]. (Click to expand)
External Quality Assessment
External Quality Assessment (EQA) or Proficiency Testing (PT) involves running blind patient-like samples and comparing your results to peer results, in order to retrospectively monitor the accuracy of reporting. EQA samples should be treated as if they were a patient sample and therefore must be run by personnel who would normally use the device. This provides confidence in the reliability of patient test results. (Learn more about EQA)
Benefits of participation in an EQA programme include assessment of result accuracy, assessment over time, comparisons with instruments, methods and peers, and providing confidence in test results.
EQA for POCT is, in theory, similar to EQA in a large laboratory. There is a significant difference however, the POCT participants are usually health care professionals with little knowledge of laboratory medicine. A lack of understanding of the importance of EQA had led to a smaller percentage of sites participating than large laboratories.
A Good EQA Scheme
A good EQA scheme should offer:
Conducting EQA in POCT
Below are some suggestions for how EQA should be conducted for POCT.
Conduct
EQA should be conducted by the operator who normally conducts patient testing to ensure the true workflow is assessed [6].
Material
EQA samples should be commutable, meaning they have the same numeric relationship between measurements procedures as is observed for a panel of patient samples (reacts the same as a real patient sample).
Report Frequency
Challenges / surveys should be frequent enough to identify systematic errors in a timely manner, affecting the fewest patient results [10].
Report Turnaround
A fast turnaround time allows test system errors to be identified sooner and necessary corrective actions to be taken immediately with minimum disruption to the lab.
Review
A regular review of past EQA results should be part of the cycle of quality.
Guidance
A POCT EQA provider should be able to provide assistance when the user is having difficulties.
Results
Individuals carrying out testing should have the correct knowledge to interpret results, choosing a scheme with easy to interpret results can help.
Internal Quality Control
Randox offer a number of controls suitable for Point of Care Testing applications:
Acusera Blood Gas Control
The Randox Acusera Blood Gas Quality Controls contain assayed target values for ten parameters, covering pH, pCO2, pO2, electrolytes, glucose and lactate. The material is provided in easy to open ampoules for added convenience and ease-of-use. The liquid ready-to-use nature of the control makes it ideal for use in point-of-care testing and on a wide range of blood gas instruments.
Acusera Liquid Cardiac Control
The Randox Acusera Liquid Cardiac control is designed to be both convenient and easy to use. The liquid ready-to-use format makes it ideal for both clinical laboratories and point-of-care testing. Assayed, instrument specific values are provided for an impressive 8 cardiac markers including, NT-ProBNP, D-dimer and Troponin ensuring consolidation and flexibility. Furthermore, an open vial stability of 30 days for all analytes helps to keep waste and costs to a minimum.
Acusera Liquid HbA1c Control
Liquid Urine Control
The Randox Acusera Liquid Urine quality control is designed to be both convenient and easy to use. The liquid ready-to-use format eliminates issues with pipetting and allows convenient storage at 2℃ – 8℃. Assayed instrument and method specific target values and ranges are provided for 18 commonly tested urine chemistry parameters.
External Quality Assessment
Randox offers RIQAS Point of Care, a simple EQA scheme designed for use in point of care settings. It is a single sample, single scheme programme featuring whole blood samples for authentic patient sample assessment.
RIQAS
RIQAS Point of Care
Acusera
[1] C. Price, A. St john and J. Hicks, “Point-of-care testing”, 2004. [Online]. Available: http://mldt.hu/upload/labor/document/PRICEP.pdf. [Accessed: 23- Jul- 2018].
[2] C. Price, “Point of care testing”, BMJ, vol. 322, pp. 1285-1288, 2001.
[3] A. Okorodudu, “Optimizing accuracy and precision for point-of-care tests”, Acutecaretesting.org, 2011. [Online]. Available: https://acutecaretesting.org/en/articles/optimizing-accuracy-and-precision-for-point-of-care-tests. [Accessed: 24- Jul- 2018].
[4] H. Holt and D. Freedman, “Internal quality control in point-of-care testing: where’s the evidence?”, Annals of Clinical Biochemistry, vol. 53, no. 2, pp. 233-239, 2016.
[5] “ISO 22870:2016 – Point-of-care testing (POCT) — Requirements for quality and competence”, Iso.org, 2018. [Online]. Available: https://www.iso.org/standard/71119.html. [Accessed: 25- Jul- 2018].
[6] J. Gill and M. Shephard, “The Conduct of Quality Control and Quality Assurance Testing for PoCT Outside the Laboratory”, Clin Biochem Rev., vol. 31, no. 3, pp. 85-88, 2010.
[7] A. Stavelin and S. Sandberg, “Essential aspects of external quality assurance for point-of-care testing”, Biochemia Medica, pp. 81-85, 2017.
[8] C. Price, I. Smith and A. Van den Bruel, “Improving the quality of point-of-care testing”, Family Practice, vol. 35, no. 4, pp. 358-364, 2017.
[9] “ISO 15189:2012 – Medical laboratories — Requirements for quality and competence”, Iso.org, 2018. [Online]. Available: https://www.iso.org/standard/56115.html. [Accessed: 31- Jul- 2018].
[10] J. Crilly, “Mythbusting: Frequency of EQA Reports”, Randox Laboratories, 2017.
[11] G. Kristensen and P. Meijer, “Interpretation of EQA results and EQA-based trouble shooting”, Biochemia Medica, pp. 49-62, 2017.
Importance of External Quality Assessment (EQA)


External Quality Assessment (EQA) / Proficiency Testing (PT) allows for a comparison of a laboratory’s testing procedures to other laboratories across the world. Comparisons can be made to a peer group of laboratories or to a reference laboratory.
EQA involves running blind patient-like samples, comparing your results to peer results, in order to retrospectively monitor the accuracy of reporting. EQA samples should be treated as if they were a patient sample and therefore must be run by personnel who would normally use the device. This provides confidence in the reliability of patient test results.
Participating in an EQA scheme allows a laboratory to gather valuable data, this data can be used in a variety of ways [1]:
EQA provides assurance to both staff and customers that testing taking place at your laboratory provides accurate and reliable results. Problems can be identified early on and corrective action can be untaken. The reliability of methods, materials, and equipment can be evaluated and training can be developed and its impact monitored.
Large laboratory groups can compare their performance with sites across their group, ensuring accuracy and consistency no matter where testing takes place.
EQA participation is often a requirement for accreditation, gaining accreditation alone has a host of benefits, not least an increased confidence in results from customers, current and potential.
Read more about accreditation: ‘The Importance of Meeting ISO 15189 Requirements’.
Point of care testing (POCT) refers to testing that is performed near or at the site of a patient with the result leading to a possible change in the care of the patient. The popularity and demand for POCT has recently seen rapid growth, this comes from the advantages including the added convenience of being able to obtain a rapid result at the patient’s bedside, thus allowing immediate action, saving time and improving the potential outcome for the patient.
Although there are many benefits of using POCT devices in terms of their convenience, these benefits are only true if the results produced are both accurate and reliable. Ensuring accuracy and reliability is the primary responsibility of Quality Control.
EQA is strongly recommended for all point of care devices and is recommended by ISO 22870, which providesspecific requirements applicable to point-of-care testing and is intended to be used in conjunction with ISO 15189.
There are many External Quality Control schemes that come in different varieties. EQA schemes can be mandatory, required either by accreditation or law. Others are voluntary and carried out by laboratories who want to ensure that they are carrying out accurate testing and improve the quality of the lab’s performance [1].
A good EQA scheme should offer:
EQA is a great tool for comparing against a peer group and maintaining an effective QC strategy, however, it has its limitations.
EQA / PT alone cannot provide a complete evaluation alone; it is important to also run third party controls regularly. You can find out about the importance of third party controls here.
EQA results can also be affected by variables not relating to patient samples, including preparation, clerical functions, matrix effects, and selection of method. The errors can appear to be a downside to EQA but it can be used as a way to evaluate staff performance as well as assay performance.
If possible, every laboratory should participate in an EQA scheme that covers all testing procedures. Laboratories need to develop a management process with the objective to assure that EQA samples are treated appropriately and in the same manner. This includes, sample handling, sample analysis, record keeping, investigating deficiencies, taking corrective actions, and communicating results with laboratory staff and management.
Problems at any stage of sample analysis can cause errors, when an error does happen, all elements of the process need to be checked. Some examples of errors:
Pre Analysis
• Incorrect sample handling during preparation, shipping or storage
• Improper storage
• The material has expired
• An error in manufacturing
Analysis
• Instrument, calibration or reagent defects
• Staff competency
• Matrix effects
• Incorrect analysis method
Post Analysis
• Report misinterpretation
• Clerical or transcription errors
• Failure to take corrective action
• Where possible, all laboratories should participate in an EQA scheme for all tests that they perform.
• EQA samples should be treated in the same way as a patient sample, using the same procedures, instruments, methods, and staff who normally perform the testing.
• EQA provides valuable resources and data to effectively maintain accurate and reliable results and should be seen as educational.
Randox offers RIQAS, the largest EQA scheme in the world with over 45,000 participants across 133 countries, offering 33 consolidated programmes. Randox also offers a range of over 90 molecular programmes for infectious disease testing with Quality Control for Molecular Diseases (QCMD).
Features and Benefits



Fully accredited to ISO/IEC 17043:2010



Consolidated programmes



High frequency reporting



The highest quality material



User-friendly reports
To learn more about RIQAS, visit the RIQAS homepage.
RIQAS
RIQAS Point of Care
QCMD
[1] WHO, Overview of External Quality Assessment (EQA). World Health Organisation, 2009.
[2] ISO 15189:2012 Medical laboratories — Requirements for quality and competence, 3rd ed. ISO, 2014.
[3] ISO 22870:2016 Point-of-care testing (POCT) — Requirements for quality and competence, 2nd ed. ISO, 2016.
RIQAS Point of Care – FAQs


RIQAS Point of Care
RIQAS
Contact Us
Liquid ready-to-use controls & their benefits
Randox Quality Control is a world leading manufacturer of third party quality controls. With an extensive product portfolio – including the largest range of liquid ready-to-use controls, Randox QC will meet your laboratory requirements and deliver trustworthy results repeatedly.
What control formats are available for your laboratory?
There are three distinct formats available for use in the laboratory with each format having benefits and drawbacks. When choosing a Quality Control material it is important you choose the most convenient solution for your laboratory requirements. The three formats are;
- Lyophilised (freeze dried)
- Liquid frozen
- Liquid stable (ready-to-use)
What format is best?
There are several possible answers to this question, simply because, every laboratory is different. What works for one laboratory may not work for another and for this reason there is no one format that works best for all laboratories.
What we can say however, is that there are varying levels of convenience across the different formats. The most convenient, and arguably, the most favoured of the three formats is liquid ready-to-use. It is not difficult to understand why this format is widely regarded as the preferred choice of control in laboratories – they are simple to use, require no preparation and can be conveniently stored and shipped at 2-8oC.
Why should you consider a liquid ready-to-use control?
There are many benefits to using a liquid ready-to-use control over both liquid frozen and lyophilised controls. One of the main benefits of a liquid stable control material is that it eliminates any potential reconstitution/pipetting errors often associated with lyophilised controls. They also eliminate the additional time taken to thaw liquid frozen controls and can significantly reduce shipping/delivery costs as they do not need to be shipped on dry ice.
Another major benefit of running a liquid ready-to-use control is the fact they are also suitable for use in Point-of-Care-Testing (POCT). With growing popularity amongst laboratory professionals and more people expecting rapid results, POCT is on the rise. Due to their easy to use nature, liquid ready-to-use controls are extremely beneficial to POCT providers.
One further benefit to using a liquid ready-to-use control is the longer open vial stability when compared to both lyophilised and liquid frozen controls. With many lyophilised and liquid frozen controls, the open vial stability can vary and generally is around 7 days when fully thawed or reconstituted in comparison to 28 days with a liquid ready-to-use control.
Randox Quality Control – Liquid Ready-to-Use Control Portfolio
The Randox QC portfolio – better known as Acusera – is convenient, hassle-free and cost effective. Our liquid ready-to-use control range includes; Liquid Cardiac, Blood Gas, Liquid Urine, Urinalysis, Specific Protein, Ammonia Ethanol, Haematology, Liquid HbA1c, Liquid CSF & Liquid Tumour Markers.
For further information on Randox Quality Control or our range of liquid ready-to-use controls please email us at acusera@randox.com.
ISO 22870- Are you meeting expectations?
Quality control has recently become crucial in the Point-of-Care (POC) field due to the introduction of ISO 22870 regulations and increased focus in patient safety. Quality control is critical in reducing turnaround time and saving money.
There is now an international standard specifically for POC testing, ISO 22870. This standard is intended to be used in conjunction with the standard for medical laboratories, ISO 15189. This means that aspects relating to Point-of-Care such as training, competence and documentation should be carefully planned, implemented and governed by a quality management system and there is a requirement for QC and EQA to be performed, where available.
POCT is typically carried out by non-laboratory staff, therefore when selecting the appropriate IQC material for POCT there are a number of key characteristics you must consider;
- Format of the material – QC material employed should be liquid stable, requiring no preparation, reducing the likelihood of human error and increasing convenience.
- Value assignment – all values must be accurately assigned. Look out for suppliers who use a large number of independent labs to determine the target value.
- Third party controls – manufactured independently from any specific instrument or method third party controls are designed to deliver unbiased performance assessment.
- Storage – for convenience controls should be liquid stable, as these can be easily stored in a fridge at 2oC – 8oC and won’t need to be shipped on dry ice.
- Stability – a control with a good open vial stability will mean that it can be used for longer with less waste produced, meaning it is more convenient for the medical professional to use.
- Transportation– the liquid stable controls can be conveniently stored at 2oC – 8oC reducing the need to ship on dry ice
- Minimal training– easy to use with little training required, therefore suitable for use by non-laboratory personnel
In addition to IQC, External Quality Assessment (EQA) must also be employed to ensure a comprehensive review of test system performance. It is best to select a programme that offers frequent reporting with a large database of users. This will enable rapid error identification and ultimately accurate and reliable patient testing.
Our Acusera liquid ready-to-use controls include:
- Blood Gas Control– A liquid stable control provided in easy to open ampoules for added convenience and ease-of-use. Assayed, method specific target values are provided for the most common blood gas instruments.
- Liquid Cardiac Control– This is a highly convenient liquid stable cardiac control offering excellent consistency. Assayed, instrument specific target values are provided for 8 cardiac markers, enabling flexibility and consolidation.
- Liquid Urinalysis Control– Liquid control that is compatible for use with both manual and automated methods of dipstick analysis. Available in convenient 12ml vials or 25ml dropper bottles with assayed ranges provided for 13 parameters covering the chemical examination of urine specimens.
- Liquid HbA1c Control– This is another highly convenient liquid ready-to-use control. With an open vial stability of 30 days, keeping waste and costs to a minimum.
Complementary EQA programmes are also available to meet the needs of ISO 22870.
ISO 22870:2006 in the Point-of-Care Testing (POCT) Market
Point-of-Care Testing (POCT)
The Point-of-Care testing market, better known as POCT, is growing rapidly as the demand for faster patient testing increases. This point is fully backed up through the results of the recent BIVDA (British IN-Vitro Diagnostics Association) survey of 2016. This survey focuses primarily on Point-of-Care Testing and there were a few questions asked that sparked an interest amongst us. The questions can be seen below;
“What are the challenges you face regarding POCT?”
“What do you see in the future for POCT?” (BIVDA, 2016)
Although many answers were put forward there was one (answer) that appeared for both questions. Accreditation. We found this intriguing yet far from surprising. POCT and accreditation go hand-in-hand, with laboratories striving to achieve ISO accreditation with the ultimate aim of allowing patients to have peace of mind that they are being tested properly and effectively.
ISO 22870:2006
The ISO standard that relates to POCT is ISO 22870:2006 Point of Care Testing- Requirements for quality and competence. This standard gives specific requirements applicable to POCT testing and apply when POCT is carried out in a hospital, clinic or a healthcare organisation providing ambulatory care. However, it is important to note and remember that ISO 22870 is not to be seen as a separate, standalone document, in fact, it is to be used in conjunction with ISO 15189:2012 (medical laboratories – particular requirements for quality & competence).
With accreditation in POCT becoming a major factor to laboratories, it is no surprise to see accreditation as one of the top answers to the two questions asked in the survey.
For laboratories looking to participate in point-of-care testing there are other factors of ISO 22870 that they must be aware of, one of the major factors is as follows;
“ISO 22870:2006 advises that where available, Internal Quality Control and participation in an External Quality Assessment (EQA) scheme is required in the point-of-care setting.”
How can Randox help?
Fortunately for laboratories, Randox are able to help out here. With a vast range of true third party controls and 32 comprehensive RIQAS (EQA) programmes, Randox can aid laboratories as they aim for ISO accreditation.
We have multiple products particularly suited to the POCT market including, Liquid Cardiac, Blood Gas and Urinalysis. These products are available both as internal quality controls and as EQA programmes.
Our Acusera range of true third party controls are conveniently supplied liquid ready-to-use, therefore requiring no preparation. This is a particular advantage in the POC environment were test results are required quickly and also were testing is not always performed by laboratory personnel. Supplied with assayed target values and manufactured from 100% human material, ensuring they act in the same manner as a patient sample, our Acusera controls are ideal for use at the point of care and will therefore aid a laboratory in gaining ISO accreditation.
Similarly, participation in our RIQAS EQA programme will also assist laboratories to meet regulatory requirements and achieve ISO accreditation. With over 40,000 laboratory participants in 123 countries, RIQAS is the largest international EQA scheme worldwide! Our Liquid Cardiac, Blood Gas and Urinalysis programmes are all supplied liquid ready-to-use, complementing their Acusera counterparts perfectly!
By using our Acusera controls and RIQAS EQA programmes side by side, a laboratory can confidently aim for ISO accreditation in the knowledge that they are meeting criteria.
Urinalysis External Quality Assessment
The RIQAS Urinalysis EQA programme is designed to monitor the performance of urine dipstick test using both manual and automated methods. All samples are supplied in a liquid ready-to-use format ideal for both clinical and point-of-care testing (POCT).
- Liquid ready-to-use
- 100% human urine
- Bi-monthly reporting
- Submit results and view reports online via RIQAS.Net
- Suitable for POCT
- Accredited to ISO/IEC 17043
| Cat No | Kit Size | Frequency | Cycle Start | Parameters | |
|---|---|---|---|---|---|
| RQ9138 | 6 x 12ml | Bi-monthly (1 x 12 month cycle) | January | 14 | |
Parameters
- Albumin
- Bilirubin
- Blood
- Creatinine
- Galactose
- Glucose
- hCG
- Ketones
- Leukocytes
- Nitrite
- pH
- Protein
- Specific Gravity
- Urobilinogen
Please note, product availability may vary country to country.
Related Products
Randox launches White Paper on Quality Control in POCT
Randox has launched the first in a series of White Papers, focusing on the importance of quality control in point-of-care testing (POCT). The White Paper is free to download and will help laboratories define an appropriate QC procedure for all POCT devices.
Patient testing at the point-of-care has increased dramatically in recent years, as technology makes it more convenient to test patient samples at the bedside producing rapid results and allowing immediate action. However, the benefits of this are only true if the results are both accurate and reliable, making quality control vitally important in the POC setting, just as it is in the laboratory.
A recent survey* identified that POCT is the largest source of error when compared to other sources of laboratory testing errors. Furthermore, the most common phase for errors in POCT is in the analytical stage, whereas in laboratory-based testing this is the least common source for errors. This highlights how vital it is to have a robust QC procedure in place for POCT as the potential risk of harm to a patient appears to be far greater for POC tests compared to those performed in a laboratory.
The White Paper from Randox covers the fundamentals of developing a POCT QC strategy in line with ISO15189 requirements, looking at who is responsible for QC, what QC is appropriate for different types of POC devices and tips for choosing IQC material and an EQA scheme.
The Paper can be downloaded from the Randox website here.
Randox second White Paper, which studies the importance of using 100% human serum controls to avoid lot-to-lot variations in immunoassay testing, will be available in December.




![Fig. A Fig. A Examples of a manual IQC documentation, adapted from the Australian Government’s POCT General Practice [4]](https://www.randox.com/wp-content/uploads/2018/08/Fig.-A.jpg)




