India Must Stop Misusing Antibiotics in Dairy Sector, According to CSE Report
India Must Stop Misusing Antibiotics in Dairy Sector, According to CSE Report
India Must Stop Misusing Antibiotics in Dairy Sector, According to CSE Report
The Centre for Science and Environment (CSE) has raised concerns over whether the milk we consume is safe. Hosting a virtual meeting in July, the CSE discussed the use of antibiotics with the world’s largest producer of milk, India. Over 188 million tonnes of milk was produced by India over the period of 2018-2019 and lead with 22% of global milk production.
A recent assessment by the CSE has found indication of antibiotic abuse in milk producing cattle.
“We have found that antibiotics are extensively misused in the dairy sector; antibiotic residues remain largely untested in milk, an integral part of Indian diets, particularly of children,” said CSE Director General Sunita Narain. “While we continue to struggle against COVID-19, we are staring at another pandemic like situation – that of antibiotic resistance fueled by the way we are producing our food, which has become chemical-intensive.”
Antibiotics such as penicillin, aminoglycosides and fluroquinolones are being ‘indiscriminately’ used by dairy farmers according to the CSE. These antibiotics are among the most important in human health. The World Health Organisation (WHO) has warned as we face growing concerns over an antibiotic resistance, such antibiotics should be preserved.
Despite laws stating that antibiotics should only be prescribed and administered by a registered veterinarian, the CSE said that some farmers are illegally attaining and injecting animals themselves without the supervision of veterinarians.
“Farmers often sell milk while the animal is under treatment, which increases the chances of antibiotic residues in the milk,” explained Amit Khurana, the CSE’s Food Safety and Toxins Programme Director. “While milk sold directly to consumers is not tested, contrary to what one would expect, processed milk sold in packets is also largely unchecked for antibiotic residues.
“This explains why, despite pooling and processing, packed milk samples from several states had antibiotic residues in the FSSAI’s [Food Safety and Standards Authority of India] milk quality survey of 2018.”
However, CSE noted the virtual meeting has encouraged positive signs of action in the diary industry in India.
“The issues highlighted by the CSE assessment have been well recognised by the experts and stakeholders. These experts have also recommended several measures for minimising antibiotic misuse in the Indian dairy sector.”
In order to minimise the threat of antibiotic resistance, the CSE have recommended “preventative measures” such as;
- Routine observation of drugs residues in milk
- Modifying the existing standards for antibiotic residues in milk
- Limiting misuse of essential antibiotics such as penicillin
Such measures, Khurana noted, are showing some success. “Information shared by various organisations, agencies and experts suggest that ethno-veterinary medicines, better management of sub-clinical mastitis, and good farm management are contributing towards reducing antibiotic misuse.”
“India is now talking about antibiotic resistance in the one-health perspective,” noted Narain. “There is a shared concern, and all voices are now together in expressing that concern.” As such he believes further (and necessary) action will be taken.
“We must ramp up surveillance and testing, put a complete stop to the use of critically important antibiotics and penalise their use,” he added, “and work with farmers and the agriculture-dairy sectors to innovate on solutions.”
Randox Food Diagnostics
Randox Food Diagnostics recognise the pressure the dairy industry is under to mass produce high quality products whilst complying with regulations and have developed the solution. A comprehensive range of easy to use, accurate milk analysis methods for the qualitative and quantitative detection of contaminants in milk.
With 6 Biochip arrays available including the revolutionary InfiniPlex for Milk, Randox Food provide the highest quality testing platforms for the screening of antimicrobials and are the only commercial supplier of a multiplexing anthelmintics array in milk.
Biochip Array Technology saves time and money whilst enabling users to get a deeper insight into milk quality and ensure a safe supply chain.
Contact us at info@randoxfooddiagnostics.com, or visit https://www.randoxfood.com/milk-analysis/ to find out more about our milk testing solutions.
Want to know more?
Contact us or visit our Randox Food Diagnostics website.
Related Products
Meat & Seafood
Milk
Honey
Wine
RX modena and Randox Reagents used in COVID-19 hospital in India
RX modena and Randox Reagents used in COVID-19 hospital in India
Randox Laboratories has installed an RX modena into a COVID-19 hospital in India; the Netaji Subhash Chandra Bose Medical College in Madhya Pradesh state.
The installation, which marks the first RX modena to be used in an Indian Government Medical College, replaces existing instrumentation in the Medical College laboratory, an initiative which was driven by College Dean Dr. Pradeep Kumar Kasar and Head of Department Dr. Ashok Kumar Sahu.
The RX modena will cater for an additional 38 samples compared to the previous instrumentation, and is running a range of biochemistry tests that play a pivotal role in COVID-19 patient management, including C-Reactive Protein (CRP), Ferritin, and Lactate Dehydrogenase (LDH).
Dr. Ashok Kumar Sahu, Head of Biochemistry at Netaji Subhash Chandra Bose Medical College, commented;
“We are very pleased with the performance of the RX modena, and in particular with its wide menu of tests that can be utilized for COVID-19 patient management. Its high throughput and versatility have been a great support for the clinicians working in our COVID-19 hospital, in determining risk stratification, disease progression, and response to treatment.”
The RX modena is a high volume, floor standing, fully automated clinical chemistry analyser with a world leading test menu which covers specific proteins, lipids, therapeutic drugs, antioxidants, diabetes and veterinary testing. The versatile analyser offers advanced methodologies with excellent correlation to the gold standard, and excellent reagent CV ranges.
Shail Dholabhai, Randox India Sales Manager, commented;
“We are delighted that the team at Netaji Subhash Chandra Bose Medical College have chosen the RX modena to fulfil the testing requirements of this COVID-19 hospital. Offering an unrivalled test menu of routine and specialized chemistries, the RX modena provides laboratories with the superior precision, reliability and accuracy that the Randox RX series is renowned for.
“Capable of performing a total of 1200 tests per hour including ISE, the RX modena presents a new class of efficiency which will result in faster and more accurate diagnoses, and ultimately, lead to better patient outcomes.”
Benefits of the RX modena
- Capable of performing 800 photometric and a total of 1200 tests per hour including ISE.
- World-leading extensive test menu covering routine chemistries, specific proteins, lipids, antioxidants, cardiac, diabetes and veterinary testing, offering cost savings through consolidation of routine and novel tests on a single platform.
- Economic platform with low water consumption of less than 20 litres per hour saving on consumable costs. The RX modena also requires less than 5 minutes minimal maintenance.
- Interactive touch-screen technology with integrated barcode readers for reagent and sample identification. Increased functionality with 13 wavelengths generated via diffraction grating (340-800nm) ensuring a multitude of chemistries are possible on one system.
- Easy-to-use advanced Microsoft 10 software with a built-in Inventory Management System calculating the number of tests remaining, providing alerts of shortages and expired reagents and calibrators. The RX modena also features automatic test re-run function and sample dilution.
- Dedicated reagent and sample pipettes to minimise the risk of errors and contamination. The RX modena also features a continuous loading hatch to allow for samples to be analysed quickly and easily during a run.
- Advanced QC capabilities with daily, monthly and batch QC with data archiving, Levey-Jennings charts and automatic QC and calibration. There are also 7 different calibration options available.
For further information about the RX modena click here.
For information about laboratory tools for COVID-19 patient management please click here.
For other enquiries please contact marketing@randox.com
Want to know more about Randox?
Contact us or visit our homepage to view more.
Our Randox Products and Services
REAGENTS
RX SERIES
ACUSERA
BIOCHIP
Antibiotic testing in shrimp
Seafood is one of the most important exports in India with its shrimp being a staple food in many countries worldwide. However, 2017 and 2018 saw 27 shipments of shrimp refused entry into the US by the FDA. This was followed closely in January 2019, when 26 lines were refused due to the presence of two banned antibiotics, nitrofurans and chloramphenicol.
With Indian shrimp accounting for around one third of the countries seafood exports, India has expressed its concern over the rejections. It responded by calling the tests on the products ‘too stringent’.
The global shrimp industry is estimated to be worth around $30 billion and India’s market share is estimated at 13% in value terms.
Dr. Ramraj, President of the All Indian Shrimp Hatcheries Association has stated, “some of the metabolites in shrimp and crustacean shells are known to mimic antibiotics and therefore could give false results”.
The use of antibiotics in shrimp farming in India is banned. Madhusudano Rao, Principal scientist at India’s Central Institute of Fisheries Technology has said, “All shrimp hatchery operators and shrimp farmers and advised to use only these antibiotic- free inputs during shrimp farming”.
Randox Food Diagnostics offer the most comprehensive range of ELISA and Biochip tests currently on the market, specifically designed to identify and detect the smallest traces of the most prevalent antibiotics used in seafood, including nitrofurans and chloramphenicol.
For more information please email us at: info@randoxfooddiagnostics.com or visit our website at www.randoxfooddiagnostics.com.
The rise of drug abuse in India – Randox Toxicology
A recent study completed by the Narcotics Control Bureau (NCB) reported an all-time high of 3.6 lakh kg narcotic drugs seized in India in 2017, this was an increase of over 300 percent in the last five years. Known as ganja, cannabis is the most widely abused drug in India, increasing from 40,113 in 1999 to 352,379 by 2017, according to The Times of India.
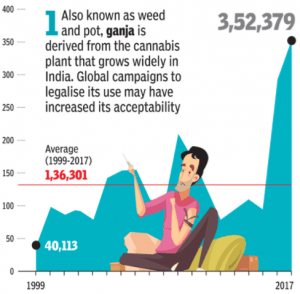
(Sourced: The Times of India: Drug abuse on the rise in India, Ganja most in demand)
A prevalent issue in the north of India is pharmaceutical drug abuse, both over the counter and prescription drugs. The Times of India reported that 566,450 bottles of cough syrup, 58,463 injections and more than 6,300,000 tablets were recovered across India in 2017. Among the drugs being smuggled out of India and into countries such as America and Canada is the opioid painkiller Tramadol, which is not covered under the Narcotic Drugs and Psychotropic Substances Act.
With daily drug seizures across the country, the situation in India has been described as akin to the US national opioid emergency. Therefore, it has been suggested that a harm-reduction approach be taken to educate the young and tackling substance abuse.
Using our revolutionary Biochip Array Technology, the Evidence MultiSTAT is a fully automated analyser that enables on-site simultaneous detection of up to 21 classical, prescription and synthetic drugs from a single sample. Designed to work across a variety of matrices and generate results in under 20 minutes, Randox Toxicology have changed the landscape of drug detection forever.
For further information about the Evidence MultiSTAT and our cutting-edge multiplex testing capabilities, contact info@randoxtoxicology.com to be put in touch with a sales member or visit www.randoxtoxicology.com
We Are Randox | Meet the team in Bangalore, India
Here at Randox, we’re a diverse bunch, spread over 145 countries in the world. We have more than 1400 employees of 44 nationalities, including 300 research scientists and engineers. Needless to say, the Randox family is a multicultural one!
We have four key manufacturing and R&D sites – in County Antrim, Northern Ireland; Dungloe, County Donegal, Ireland; Bangalore, India; and the Greater Washington DC area, in the U.S. This month, our We Are Randox article focuses on the team in Bangalore.
Randox India, located alongside other high-tech industries based in Bangalore, consists of 37 office staff and 77 field staff, including customer support engineers and sales managers. A base for administration, sales and manufacturing in India, the Bangalore site, set in the capital of the Indian state of Karnataka, services the 3.3 million square kilometre country.
Photographed are members of the Randox team in Bangalore from the following departments;
- Accounts and Finance
- Customer Support Engineers
- Logistics and Trading
- Manufacturing
- Quality Control
- Research & Development
- Administration
- Travel
- Human Resources
Brian Walsh, Manufacturing Manager at Randox India, said:
“The photograph below was taken during this year’s Diwali, which is the Hindu festival of lights celebrated every year in autumn in the northern hemisphere. Danny Maguire, who is based in Ardmore at Randox HQ, was over on business at the time and joined us in celebrating this cultural tradition.
“It is one of the most popular festivals of Hinduism, and spiritually signifies the victory of light over darkness, good over evil, knowledge over ignorance, and hope over despair.
“We all really enjoyed having Danny with us and sharing some of our customs and values with him, many of which our team members based in other sites across Randox would not know about.
“We hope to welcome many more of our colleagues from across the globe to Randox India in the near future!”

Want to know what it’s like to work in Bangalore? Read all about when we met up with Pankaj Chitkara, who is our National Sales Manager for the RX Series in India.
For further information on the Randox Bangalore team, please contact the Randox PR team via email: randoxpr@randox.com or phone 028 9442 2413


















