The rise of drug abuse in India – Randox Toxicology
A recent study completed by the Narcotics Control Bureau (NCB) reported an all-time high of 3.6 lakh kg narcotic drugs seized in India in 2017, this was an increase of over 300 percent in the last five years. Known as ganja, cannabis is the most widely abused drug in India, increasing from 40,113 in 1999 to 352,379 by 2017, according to The Times of India.
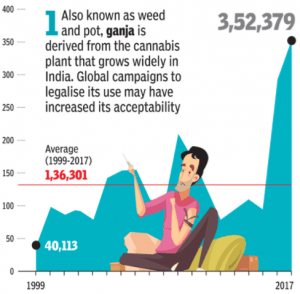
(Sourced: The Times of India: Drug abuse on the rise in India, Ganja most in demand)
A prevalent issue in the north of India is pharmaceutical drug abuse, both over the counter and prescription drugs. The Times of India reported that 566,450 bottles of cough syrup, 58,463 injections and more than 6,300,000 tablets were recovered across India in 2017. Among the drugs being smuggled out of India and into countries such as America and Canada is the opioid painkiller Tramadol, which is not covered under the Narcotic Drugs and Psychotropic Substances Act.
With daily drug seizures across the country, the situation in India has been described as akin to the US national opioid emergency. Therefore, it has been suggested that a harm-reduction approach be taken to educate the young and tackling substance abuse.
Using our revolutionary Biochip Array Technology, the Evidence MultiSTAT is a fully automated analyser that enables on-site simultaneous detection of up to 21 classical, prescription and synthetic drugs from a single sample. Designed to work across a variety of matrices and generate results in under 20 minutes, Randox Toxicology have changed the landscape of drug detection forever.
For further information about the Evidence MultiSTAT and our cutting-edge multiplex testing capabilities, contact info@randoxtoxicology.com to be put in touch with a sales member or visit www.randoxtoxicology.com
Biochip Carrier Applications in the Randox Evidence Series
Biochip Array Technology is the fundamental component which drives the Randox Evidence Series immunoanalysers. The simultaneous detection of multiple analytes from a single sample allows for a complete patient profile, providing exceptional time, resource and cost savings.
The Evidence Series analysers all carry the Biochip in different applications. In order to gain a better understanding of how it is utilised, the illustrations below demonstrate how adaptable and flexible the technology is across the Evidence Series.
Evidence Evolution

- One strip contains 10 biochips
- Analyser can hold 20 strips
- Can produce a throughput of up to 2640 tests per hour
Evidence

- Each cassette contains 10 carriers
- Each carrier has 9 wells
- Analyser can hold 4 cassettes at once
- Can produce a throughput of up to 3960 tests per hour
Evidence Investigator

- Analyser holds 1 cartridge
- 1 cartridge contains 9 wells
- Thermoshaker capable of carrying 6 cartridges
- Can produce a throughput of up to 2376 tests per hour
Evidence MultiSTAT

- Analyser holds 1 cartridge
- Cartridge comes pre-loaded with reagents and QC
- Can produce a throughput of up to 132 tests per hour
About the Randox Evidence Series
The Evidence Series is set to revolutionise diagnostic testing forever. Offering unrivalled capabilities across all analysers, we truly believe that the Evidence Series range of immunoassay analysers can meet your diagnostic testing capabilities.
For more information on any of the Evidence Series analysers, visit https://www.randox.com/evidence-series/ or contact us evidenceseries@randox.com.
We are attending Bee Happy Bulgaria 2018 on 1st – 4th March!
Bees and other pollinators are vital to three-quarters of the world’s food crops but have been in serious decline in recent decades. The destruction of wild habitats, disease and widespread pesticide use are all important factors.
Global honey production is projected to reach 2.4 million tons by 2022 this is driven by a growth in consumers demand for a natural and healthy alternative to artificial sweeteners, a rising awareness over the benefits of using honey as a sweetener over cane sugar and the realisation of the antibacterial and inflammatory properties of manuka honey.
The decline in the numbers of bees and rising demand from consumers for natural honey products has created a market for “illegal honey” being produced to supply the demand. Producers ‘cut’ their honey with additives like high-fructose corn syrup and brand it as natural honey, affecting purity and reducing cost.
‘Honey laundering’ has also become an issue, with countries such as China using non FDA approved chemicals within beekeeping and being non-compliant to regulations causing their honey to be banned from entering the USA. To get around this, Chinese honey is being shipped into countries such as Vietnam where it is then re labelled and moved into the USA with a new country of origin on the label. This has created the need for a reliable and accurate way to test honey globally.
In order to help the industry Randox Food Diagnostics developed the Evidence Investigator analyser. The Investigator uses Biochip Array Technology (BAT), a technology that was developed by Randox, to detect multiple residues (up to 45) from a single sample. Tests for honey include a vast range of antimicrobials such as Quinolones, Streptomycin, Chloramphenicol, Nitrofurans and more. Randox Food Diagnostics are also releasing a pesticides array in early 2018. Also offered are a range of ELISA test kits.
Alongside the evidence Investigator Randox Food Diagnostics developed the RX misano for enzymatic analysis of honey. The RX misano is now available for the analysis of diastase, total sugars (glucose/fructose), HMF (hydroxymethylfurfural) & colouration, with sucrose coming in 2018.
Randox Food Diagnostics will be attending Bee Happy Expo in Bulgaria from 1st – 4th March 2018 Stop by booth 25 or catch our presentation on Saturday 3rd March at 14:30 to learn more about our product offering for honey.
For more information visit the Randox Food Diagnostics website at: http://www.randoxfood.com/matrices/honey
Or email: info@randoxfooddiagnostics.com
Evidence MultiSTAT: Fast, Fully Automated and Versatile
The Evidence MultiSTAT is a fully automated immunoanalyser that enables on-site simultaneous detection of up to 44 analytes from a single sample of oral fluid, urine or blood. It requires a small sample volume and generates results in less than 20 minutes. The process from sample entry to results can be completed in 3 simple steps, with minimum room for human error.
The MultiSTAT offers a very simple drug screening solution to those who have no knowledge of laboratory procedures through combining simplicity with cutting-edge innovation. It is this innovative technology that enables the MultiSTAT to be easy to use, yet extremely powerful.
Versatility
Whether testing is needed for new entrants going into prison, or for a drug drive test – the MultiSTAT is adaptable for use across a range of different applications. As an analyser, it can deliver highly accurate and reliable laboratory quality results. Its ability to provide these quickly and over a wide range of substances makes it as flexible as any point of care test. The Evidence MultiSTATs ease of use makes it accessible to all with no previous laboratory experience required.
The Evidence MultiSTAT is a versatile analyser that provides automated drugs screening in a range of settings, such as low throughput laboratories, workplaces and police stations. Using chemiluminescence as a measurement principle, the Evidence MultiSTAT consistently delivers accurate results and offers a highly sensitive way to detect drugs of abuse.
Extensive
The MultiSTAT facilitates on-site simultaneous screening of multiple drug classes, including classical, prescription and synthetic drugs of abuse. Available matrices include oral fluid, with blood and urine coming soon.
Recently, the InfiniPlex for Milk Array has been developed by Randox Food Diagnostics to help the dairy industry improve the global standards of dairy. Complying with 98% of EU regulated antibiotics, InfiniPlex is currently unrivalled in the market with the ability to simultaneously detect anti-parasitics, anti-inflammatories and unauthorised substances found in dairy products from a single sample of raw milk using Biochip Array Technology. With results screened in just under 20 minutes, the Evidence MultiSTAT is ideally suited to the fast-paced nature of the dairy industry.
Developed with the user in mind, no technical expertise is required to use the test and is available on two analysers in the Evidence Series, the Evidence MultiSTAT and Evidence Investigator.
About the Randox Evidence Series
The Evidence Series is set to revolutionise diagnostic testing forever. Offering unrivalled capabilities across all analysers, we truly believe that the Evidence Series range of immunoassay analysers can meet your diagnostic testing capabilities.
For more information on the Evidence MultiSTAT, or any of the Evidence Series analysers, visit https://www.randox.com/evidence-series/ or contact us evidenceseries@randox.com.
Powering the Evidence Series – Biochip Array Technology
In 2002, Randox invented a worlds first; Biochip Array Technology, instantly changing the landscape of diagnostic testing forever. Biochip Array Technology is a multi-analyte platform which provides an unrivalled increase in patient information per sample. Instead of a patient sample needing to be subdivided for each test result, or in some cases re-collected, Biochip Array Technology offers a diagnostic patient profile with each patient sample.
How does it work?
Biochip Array Technology is a precision multiplex testing platform allowing for the simultaneous quantitative or qualitative detection of a wide range of analytes from a single sample.
The biochip detection system is based on a chemiluminescent reaction. This is the emission of light, without heat, as a result of a chemical reaction. An enzyme is used to catalyse the chemical reaction on the biochip which generates the chemiluminescent signal. The light emitted from the chemiluminescent reaction that takes place in each DTR is simultaneously detected and quantified using a Charge-Coupled Device (CCD) Camera.
Each biochip has up to 49 Discrete Test Regions (DTR). This means that up to 44 tests can be carried out simultaneously. The additional DTR are reserved for internal quality control and visual reference, a unique Biochip Array Technology feature.
How is the technology applied?
With over £250 million invested into Biochip Array Technology research and development, Randox have launched a range of Biochip Array Technology immunoanalysers – The Evidence Series. This includes the Evidence, the Evidence Evolution, the Evidence Investigator and the Evidence MultiSTAT. Each analyser is developed with boundary pushing engineering, designed to make financial, labour and time savings for the end user.
The Evidence Series has truly revolutionised diagnostic testing forever. Offering unrivalled capabilities across all analysers, we truly believe that the Evidence Series range of immunoassay analysers can meet your diagnostic testing capabilities.
For more information on any of the Evidence Series, please visit http://www.randox.com/evidence-series/ or contact us evidenceseries@randox.com.
The Evidence Evolution: The world’s most advanced immunoassay analyser
What sets the Evidence Series apart from its competitors is its flexibility across the range of immunoassay analysers. No other range of analysers has the capability in terms of throughput in comparison to the Evidence Series. With this established, we are delighted to introduce the latest in the Evidence Series range, the Evidence Evolution.
Join the Evolution
We believe that the Evidence Evolution is the world’s most advanced immunoassay analyser. The Evidence Evolution is the most versatile analyser Randox has ever produced. By utilising the same multiplex technology as the other Evidence Series analysers, the Evidence Evolution can process up to 44 results from a single sample, with a maximum throughput of up to 2640 tests per hour. With such a high volume of potential throughput, you may think that reporting time will be compromised, that is simply not the case. The Evidence Evolution can produce the first set of results in less than 36 minutes, with one sample of up to 44 results reported every minute after. The speed of reporting time distinguishes the Evidence Evolution from its competitors, and allows the analyser to further display its capabilities.
Innovation
By utilising patented Biochip Array Technology, the Evidence Evolution offers batch analysis, true random access and STAT testing capabilities. These, combined with retrospective testing, allows the user to build a more in-depth patient profile.
With such swift reporting times, the Evidence Evolution can boast of a true walkaway time of 2 hours, in a laboratory setting, this can be vital. With multiple daily tasks to be completed, time is of the essence, and the Evidence Evolution has been designed to lessen the workload of laboratory staff. By being able to offer a combination of continuous sample and reagent loading, plus automated on-board sample dilution, the Evidence Evolution will have a positive impact in busy laboratories.
Complete Picture
A fast reporting time and high throughput capabilities is just the tip of what the Evidence Evolution can offer. The analyser has the capability to test across multiple matrices, allowing for more flexibility in sampling. It also comes equipped with state of the art intuitive software and advanced precision bio-drive robotics, meaning the Evidence Evolution is an analyser that has been designed and developed to meet the needs of a modern, high throughput laboratory, by a company that has a deep understanding of laboratory needs.
About the Randox Evidence Series
The Evidence Series is set to revolutionise diagnostic testing forever. Offering unrivalled capabilities across all analysers, we truly believe that the Evidence Series range of immunoassay analysers can meet your diagnostic testing capabilities.
For more information on the Evidence Evolution, or any of the Evidence Series analysers, please visit http://www.randox.com/evidence-series/ or contact us evidenceseries@randox.com.
From Columbia to the UK: The trail that’s fuelling the worldwide cocaine epidemic
With the demand for cocaine in the UK rocketing, Top British Chef Gordon Ramsay has recently travelled to Columbia to the source of the problem, to learn more about this growing epidemic.
Having spent time with local farmers who own cocaine plants that resemble vineyards, and visiting Honduras, one of the most dangerous countries and known as the world’s warehouse of cocaine, he emphasized the urgent need to combat the drugs industry that is consuming this country.
Highlighted throughout the two-part series was the amount of cocaine that is being trafficked to the shores of the UK each year. Speaking with the metropolitan police who tackle up to 200 drug gangs in London alone, it was estimated that up to 300 tons of cocaine, with a street value of £10 billion, is being brought in to the country through a web of secretive routes used by drug traffickers.
Gordon commented that not only is cocaine a growing global problem, but it is one that is prevalent within the culinary world, including his own restaurants where he has been asked to dust the drug on top of soufflés as icing sugar, and found traces of this deadly drug in both his staff and customer toilets.
With the cocaine problem showing no signs of slowing down, Randox Toxicology are constantly working to develop the newest drug tests to meet the demands of the ever-growing drug market. The Evidence MultiSTAT was developed for use in non-laboratory settings such as workplaces, police stations and low throughput laboratories and to provide highly accurate qualitative as quickly as possible.
This revolutionary analyser enables on-site simultaneous detection of up to 21 classical, prescription and synthetic drugs from a single patient sample, reducing the burden on current systems, the risk of adulteration and helping the fight against the cartels who control the cocaine trade in South America.
For more information on our Evidence Analysers or any of our drug panels email info@randoxtoxicology.com
Welcome to the Evidence Series
Introduction
For over 30 years, we have been researching and manufacturing market-leading diagnostics products globally. With a mission statement of ‘dedicated to improving health worldwide’ the patient needs are a central focus of everything we do. It is this experience and commitment to improving healthcare that has allowed us to continually improve our product offering and make advancements globally to reach as many people as possible and provide greater diagnostic facilities.
From this experience and commitment to research, we believe we have developed a technology that has changed diagnostic testing forever.
Biochip Array Technology
In 2002, we invented a world first, which changed the landscape of diagnostic testing forever. Biochip Array Technology is a precision multiplex testing platform allowing for the simultaneous quantitative or qualitative detection of a wide range of analytes from a single sample.
Biochip Array Technology uniquely offers immunoassay diagnostic testing for simultaneous multi-analyte biomarker detection. After addition of a single patient sample to the biochip, analytes present in the sample bind to the specific biochip bound ligands. The degree of binding is determined using a chemiluminescent light source and quantified using a Charge Coupled Device (CCD) camera and imaging system.
An individual biochip has up to 49 Discrete Test Regions with each detecting a different biomarker. That means up to 44 tests can be carried out simultaneously, with the additional DTRs being reserved for visual quality control and visual reference; a feature unique to Biochip Array Technology.
The Evidence Series
Having developed this patented technology following £250 million investment in research and development, we needed a platform that allowed Biochip Array Technology capabilities to be showcased. Step forward the Evidence Series. The series includes the Evidence, the Evidence Evolution, the Evidence Investigator and the Evidence MultiSTAT. Each analyser has been designed and built with boundary pushing engineering, to ensure financial, labour and time savings for the end user.
Have a read below of the brief overview of each analyser in the Evidence Series
Evidence Evolution
The world’s first fully automated random-access biochip testing platform, the Evidence Evolution is the world’s most advanced immunoanalyser. With the capability to process up to 2,640 tests per hour, the Evidence Evolution offers complete system integration, as well as the most comprehensive test menu on the market.
Evidence
As the world’s first Biochip Array Technology system, the Evidence immunoanalyser has revolutionised laboratory screening worldwide. With the capability to process 3,960 tests per hour and a sample capacity of 360, the Evidence is ideal for use in a high throughput laboratory.
Evidence Investigator
The Evidence Investigator is a compact, semi-automated benchtop immunoanalyser that offers efficient and comprehensive testing across a range of applications including clinical diagnostics, molecular, toxicology and food diagnostics. The Evidence Investigator boasts a throughput of up to 2,376 tests per hour, offering efficiency without compromising on accuracy.
Evidence MultiSTAT
The Evidence MultiSTAT is a fully automated immunoanalyser that enables on-site simultaneous detection of up to 44 analytes from a single sample of oral fluid, urine or blood. With a three-step process and results generated in less than 20 minutes, the Evidence MultiSTAT is an ideal solution for those with no knowledge of laboratory procedures and offers a throughput of up to 132 tests per hour.
About the Randox Evidence Series
The Evidence Series is set to revolutionise diagnostic testing forever. Offering unrivalled capabilities across all analysers, we truly believe that the Evidence Series range of immunoassay analysers can meet your diagnostic testing capabilities. For more information on any of the Evidence Series analysers, please visit http://www.randox.com/evidence-series/ or contact us evidenceseries@randox.com.
Randox launching new product range at leading laboratory medicine event in USA
A group of 30 delegates from global healthcare company Randox Laboratories has this week travelled to the USA, to take part in the world’s largest diagnostics conference – in San Diego, California.
The American Association of Clinical Chemistry (AACC) Annual Meeting and Clinical Lab Expo, known as the leading event for laboratory medicine worldwide, is attended by over 20,000 delegates from across the globe, and offers Randox the opportunity to showcase their capabilities and to network with leading healthcare professionals and key decision makers.
Launching at this year’s event are a number of innovative and exciting new products, including a test for Acute Kidney Injury, a revolutionary diagnostic for small, dense low density lipoprotein (sLDL), a subtype of LDL cholesterol which is a vital risk marker for cardiovascular disease, and the latest in laboratory quality assurance software.
Chief Scientist at Randox Laboratories, John Lamont, who is heading up the delegation to the US, commented;
“Our very significant investment in research and development means that we have more new tests in development than any other healthcare company in the world. Each year at AACC we are able to bring a wealth of exciting new technologies to the American market, for a range of the world’s pressing health issues in need of the most urgent address. We look forward to showcasing our latest innovations at this year’s AACC conference, and to identifying further potential projects.”
Dr. Peter FitzGerald, Founder and Managing Director of Randox Laboratories, commented;
“We appreciate that post-Brexit there will of course be particular business challenges with regards to international business, but at Randox we will remain committed to developing new health diagnostic technologies in the areas where they are needed most, and to expanding the business in our key markets, such as the US.
“The USA is one of our most important markets and we have been exporting our diagnostic products there since the early 1980s. We will continue to nurture our presence there, the expansion of which will be supported by our soon to be opened facility in Kearneysville, West Virginia, which will enable us to strengthen our position in that market.”
AACC runs from the 30th July – 3rd August at the San Diego Convention Centre. Randox can be found at booth #3839.
For further information about Randox at AACC please contact Randox PR on 028 9445 1016 or email RandoxPR@randox.com
Evidence Series Immunoanalysers

Powered by Biochip Array Technology
In 2002, Randox invented a world first, Biochip Array Technology (BAT), instantly changing the landscape of diagnostic testing forever. BAT is a multi-analyte platform which provides an unrivaled increase in patient information per sample. Instead of a patient sample needing to be subdivided for each test result, or in some cases re-collected, Biochip Array Technology offers a diagnostic patient profile with each patient sample. So now the patient’s needs become the focus, as BAT delivers the multiple results needed for improved diagnosis.
With over £250 million invested into Biochip Array Technology research and development, Randox have launched a range of Biochip Array Technology immunoanalysers – The Evidence Series. This includes the Evidence, the Evidence Investigator and the Evidence MultiSTAT. Each analyser is developed with boundary pushing engineering, designed to make financial, labour and time savings for the end user. Utilising this technology, the Evidence series guarantees cost-effective, highly accurate and flexible testing solutions.
Why choose the Evidence Series?
Biochip Array Technology Test Menu
Adhesion Molecules
| E-Selectin | P-Selectin | L-Selectin | |
| Intercellular Adhesion Molecule-I – ICAM-I | Vascular Cell Adhesion Molecule-I –VCAM-I | ||
Alzheimer’s
| Apolipoprotein E4 –ApoE4 | Pan Apolipoprotein E – Apo E |
Anaemia
| Ferritin | Folate | Vitamin B12 |
Bone Disease
| Vitamin D | |||
Cancer
| Carcinoembryonic Antigen – CEA | Free Prostate Specific Antigen − FPSA | Total Prostate Specific Antigen − TPSA | |
Cardiac
| Cardiac Troponin I – cTnl | Creatine Kinase MB – CKMB | Heart Fatty Acid Binding Protein – H-FABP | Myoglobin |
Cerebral
| Brain-Derived Neurotrophic Factor − BDNF | Neuron Specific Enolase − NSE | ||
Cytokines
| Epidermal Growth Factor − EGF | Granulocyte Macrophage Colony Stimulating Factor | Interferon-γ − IFN-γ | Interleukin-1 alpha − IL-1α |
| Interleukin-1 beta − IL-1β | Interleukin-2 − IL-2 | Interleukin-3 − IL-3 | Interleukin-4 − IL-4 |
| Interleukin-5 − IL 5 | Interleukin-6 − IL-6 | Interleukin-7 − IL-7 | Interleukin-8 − IL-8 |
| Interleukin-4 − IL-4 | Interleukin-5 − IL 5 | Interleukin-6 − IL-6 | Interleukin-7 − IL-7 |
| Interleukin-8 − IL-8 | Interleukin-10 − IL-10 | Interleukin-12p70 − IL-12p70 | Interleukin-13 − IL-13 |
| Interleukin-15 − IL 15 | Interleukin-23 − IL-23 | Macrophage Infl ammatory Protein-1α − MIP-1α | Matrix Metalloproteinase 9 − MMP 9 |
| Monocyte Chemotactic Protein-1 − MCP-1 | Soluble IL-2 Receptor Alpha − sIL-2Rα | Soluble IL-6 Receptor − sIL-6R | Soluble Tumour Necrosis Factor Receptor 1 − sTNFR1 |
| Soluble Tumour Necrosis Factor Receptor 2 − sTNFR2 | Tumour Necrosis Factor-α − TNF-α | Vascular Endothelial Growth Factor − VEGF | |
Diabetes
| Insulin | |||
Endocrine
| Cortisol | Dehydroepiandrosterone-Sulphate- DHEAS | ||
Fertility / Pregnancy
| Estradiol | Follicle Stimulating Hormone − FSH | Luteinizing Hormone − LH | Progesterone |
| Prolactin | Sex Hormone Binding Globulin − SHBG | Testosterone | |
Fibrinolysis
| D-Dimer |
Gastro
| Gastrin 17 – GI7 | Helicobacter pylori – H. pylori | Pepsinogen I – PGI | Pepsinogen II – PGII |
Metabolic
| Adiponectin | Ferritin | Insulin | Leptin |
| Plasminogen Activator Inhibitor − PAI-1 | Resistin |
Renal
| Adiponectin | Complement C3a des Arginine – C3a des Arg | CRP (C-Reactive Protein) | Cystatin C |
| D-Dimer | Epidermal Growth Factor − EGF | Fatty Acid Binding Protein-1 − FABP1 | Interleukin-8 − IL-8 |
| Macrophage Infl ammatory Protein-1α − MIP-1α | Neutrophil Gelatinase – Associated Lipocalin – NGAL | Soluble Tumour Necrosis Factor Receptor 1 − sTNFR1 | Soluble Tumour Necrosis Factor Receptor 2 − sTNFR2 |
Stroke
| Brain-Derived Neurotrophic Factor − BDNF | D-Dimer | Glial Fibrillary Acidic Protein − GFAP | Glutathione S – Transferase Pi – GSTPi |
| Heart Fatty Acid Binding Protein – H-FABP | Interleukin-6 − IL-6 | Nucleoside Diphosphate Kinase – NDKA | Neuron Specifi c Enolase − NSE |
| Parkinson Protein 7 − PARK-7 | Soluble Tumour Necrosis Factor Receptor 1 − sTNFR1 | ||
Thyroid
| Anti-Thyroglobulin − Anti-Tg | Anti-Thyroid Peroxidase − Anti-TPO | Free Tri-iodothyronine − FT3 | Free Thyroxine − FT4 |
| Thyroid Stimulating Hormone − TSH | Thyroxine Binding Globulin − TBG | Total Tri-iodothyronine − TT3 | Total Thyroxine − TT4 |
Toxicology
| Amphetamine | Barbiturates | Benzodiazepines I | Benzodiazepines II |
| Buprenorphine | Cannabinoids – THC | Cocaine Metabolite | Dextromethorphan |
| Fentanyl | Ketamine | Meprobamate | Methadone |
| Opiate | Oxycodone I | Oxycodone II | Phencyclidine – PCP |
| Tramadol | Tricyclic Antidepressants | Zolpidem | |
Molecular
| 20 SNPs | Adenovirus A/B/C/D/E | APOB – 1 mutation | Bordetella pertussis |
| BRAF – 1 mutation | Chlamydia trachomatis – (CT) | Chlamydophila pneumoniae | Coronavirus 229E/NL63 |
| Coronavirus OC43/HKU1 | Enterovirus A/B/C | Haemophilus ducreyi – (HD) | Haemophilus influenzae |
| Herpes simplex Virus 1– (HSV-1) | Herpes simplex Virus 2 – (HSV-2) | Human Bocavirus 1/2/3 | Human Metapneumovirus – hMPV |
| Influenza A/B | KRAS – 16 mutations | LDLR – 38 mutations | Legionella pneumophila |
| Moraxella catarrhalis | Mycoplasma genitalium – (MG) | Mycoplasma hominis – (MH) | Mycoplasma pneumoniae |
| Neisseria gonorrhoea – (NG) | Parainfluenza Virus 1/2/3/4 | PCSK9 – 1 mutation | PIK3CA – 3 mutations |
| Respiratory Syncytial Virus a – RSVa | Respiratory Syncytial Virus b – RSVb | Rhinovirus A/B | Streptococcus pneumoniae |
| Treponema pallidum – (TP) | Trichomonas vaginalis – (TV) | Ureaplasma urealyticum – (UU) | |
Veterinary Residues / Food Diagnostics
| 17β-Clostebol | 5-hydroxy Flunixin | Aflatoxin B1 | Aflatoxin G1/G2 |
| Aflatoxin M1 | AHD | Amikacin/Kanamycin | Amino Benzimidazoles |
| Amoxicillin | AMOZ | Amphenicols | Ampicillin |
| AOZ | Apramycin | Avermectins | Bacitracin |
| Baquiloprim | Benzimidazoles | Beta-agonists | Beta-Lactams |
| Boldenone | Cefapirin | Cefoperazone | Cefquinome |
| Ceftiofur | Cefuroxime | Cephalexin | Cephalonium |
| Chloramphenicol | Chlormadinone | Clopidol | Cloxacillin |
| Corticosteroids | Dapsone | Decoquinate | Deoxynivalenol |
| Dexamethasone | Diacetoxyscirpenol | Diclazuril | Dicloxacillin |
| Dihydrostreptomycin | Ergot Alkaloids | Erythromycin | Ethinylestradiol |
| Fumonisins | Gentamicin | Gestagens | Halofugine |
| Hygromycin B | Imidocarb | Kanamycin | Lasalocid |
| Levamisole | Lincomycin | Lincosamides | MaduramicinG |
| Melamine | Meloxicam | Metamizole | Methyltestosterone |
| Monensin | Moxidectin (MXD) | Nandrolone | Neomycin/Paromomycin |
| Nicarbazin | Nitroimidazoles | Nitroxynil | Novobiocin |
| Ochratoxin A | Oxacillin | Paxilline | Penicillin G |
| Penicillin V | Phenylbutazone | Pirlimycin | Polymixins |
| Quinolones | Ractopamine | Rifaximin | Robenidine |
| Salinomycin | SEM | Spectinomycin | Spiramycin |
| Spiramycin/Josamycin | Stanozolol | Stilbenes | Streptomycin |
| Sulfaguanidine | Sulfamethazine | Sulphachlorpyridazine | Sulphadiazine |
| Sulphadimethoxine | Sulphadoxine | Sulphamerazine | Sulphamethazine |
| Sulphamethizole | Sulphamethoxazole | Sulphamethoxypyridazine | Sulphapyridine |
| Sulphaquinoxaline | Sulphathiazole | Sulphisoxazole | Sulphonamides |
| T2 toxin | Tetracyclines | Thiabendazole | Thiamphenicol |
| Tobramycin | Tolfenamic Acid | Toltrazuril | Trenbolone |
| Triclabendazole | Trimethoprim | Tylosin | Tylosin B/Tilmicosin |
| Virginiamycin | Virginiamycin M1 | Zearalenone | Zeranol |





























